Lidia Chitimia-Dobler
Key points
- The natural cycle of the TBE virus is dependent on vector ticks and reservoir hosts.
- There are differing transmission cycles in varying environments, from cold northern coniferous forests to temperate central European forests.
- Within a natural transmission cycle there are different ways of transmission – tick-to-tick (transovarial, sexual), host-to-tick (viremic), and also tick-to-tick and host-to-host.
- The complexity of natural transmission cycles is inadequately explored and poorly understood.
Introduction
Ticks play a critical role in the transmission of a wide variety of viral, bacterial, and protozoan pathogens to humans and animals.1,2 In the case of humans, infection is accidental as these transmission cycles are invariably enzootic with the natural hosts most frequently being wild birds and mammals.1 In order to be tangentially affected by such cycles, humans must be bitten by a vector tick species found in habitats visited by humans, as well as the tick’s usual hosts, as the dispersal of ticks not attached to hosts covers only very short distances. In addition, the tick has to accept humans as a suitable host, meaning that the species involved usually have a broad host spectrum.
Nevertheless, these tick species may only be part of the transmission cycle, with eco-epidemiologically significant sub-cycles involving tick species not commonly in contact with humans.3,4 Thus, the transmission of tick-borne pathogens often comprises a complex network of interactions involving several tick and host species. Below, we provide background to the biology of ticks and how this can influence, specifically, the eco-epidemiological cycle of tick-borne encephalitis virus (TBEV).
Structure and morphology
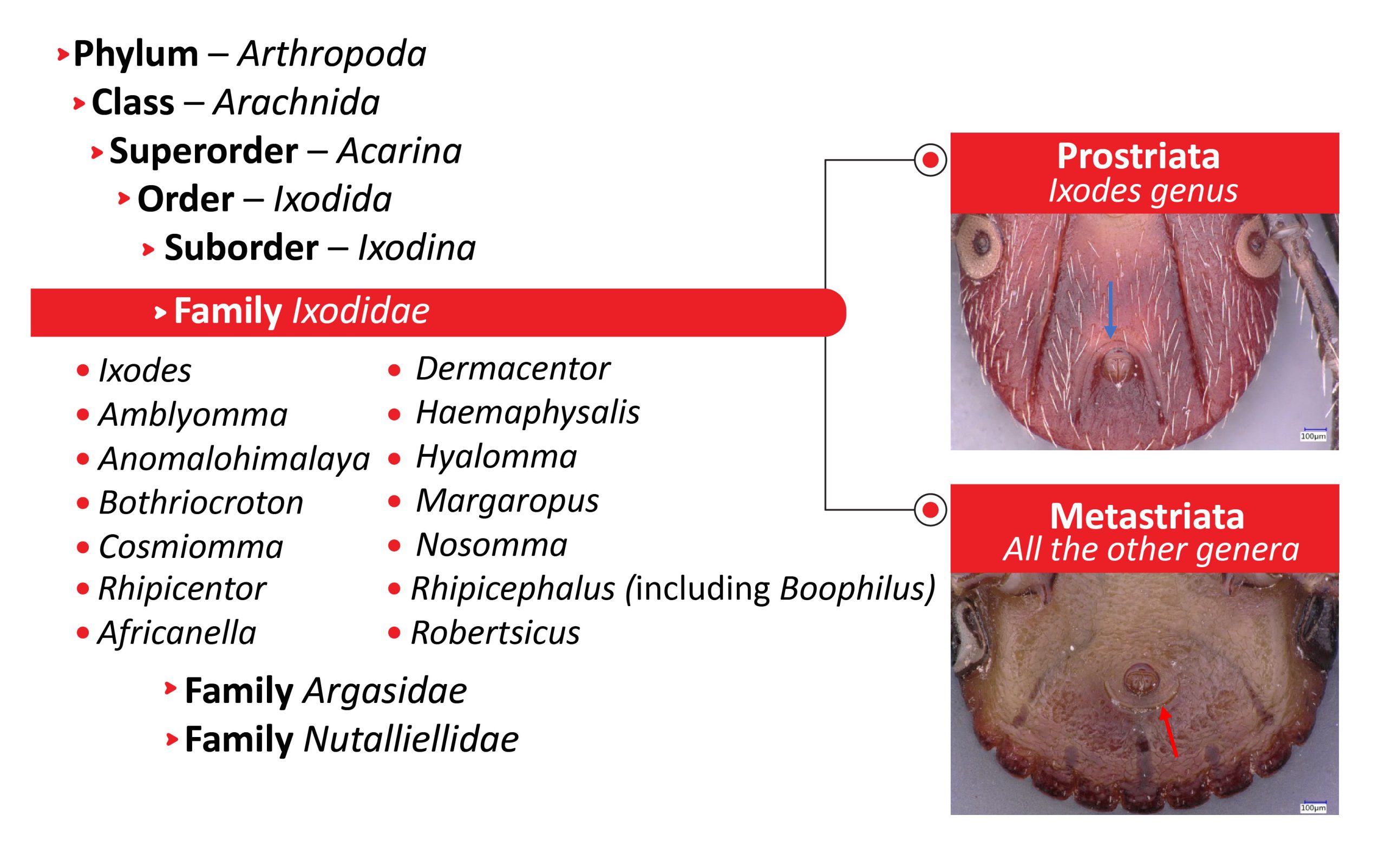
Ticks are a group of hematophagous ectoparasites with about 989 living species.5 They belong to the phylum Arthropoda, the class Arachnida, the superorder Acarina, and the order Ixodida, and they are exclusively parasitic. The Ixodida contain 3 families: the Ixodidae with 15 genera (hard ticks), the Argasidae with 15 genera (soft ticks), and the Nuttalliellidae, represented by only one species, Nuttalliella namaqua.6-9, A summary of tick taxonomy and the two groups is presented in Figure 1. All the tick species involved in the eco-epidemiological cycle of TBEV belong to the Ixodidae. Details of tick biology generally can be found in a variety of publications, for example in Nicholson et al.,9 Petney et al.,10 and Sonenshine and Roe,11 and a list of valid species names in Guglielmone and Nava.12 The following genera of ticks contain species known to transmit TBEV or in their species TBEV was detected.
Figure 1: The Ixodidae family
The Ixodidae family is divided in two groups: Prostriata, which includes only the genus Ixodes and which is characterizad by an anal groove encircling the anus anteriorly (blue arrow); and Metastriata, including 14 genera, which all have an anal groove behind the anus (red arrow).
Click the image above to enlarge
Ixodes is the largest tick genus, with 266 described species worldwide.5 Ixodes species are characterized by a distinct groove that encircles the anus anteriorly and a lack of eyes. Males have seven sclerotized ventral plates that are absent in the males of other genera. The genus Ixodes has been subdivided in roughly 15 subgenera (e.g., Ixodes, Pholeoixodes) on the basis of morphology.13,14 The genus has a worldwide distribution, including parts of Antarctica.9 Some species are particularly important as vectors of TBEV: Ixodes ricinus the castor bean tick or sheep tick in Europe and middle Asia, Ixodes persulcatus the taiga tick in northeastern Europe and northern Asia, and Ixodes ovatus in the forest belt of middle Asia and Japan.
The genus Dermacentor has 44 species worldwide.9 The basis capitulum appears rectangular when viewed dorsally. A pair of medially directed spurs occurs on the first pair of coxae. The palps are short and thick. The scutum is almost always ornamented, eyes present. Dermacentor species are found mostly in Europe, Asia, and North America.[9] In Europe, TBEV has been recovered from 2 species, Dermacentor reticulatus (the ornate dog tick), Dermacentor marginatus (the ornate sheep tick), and in Asia from Dermacentor nuttalli.
Haemaphysalis Haemaphysalis is the second largest (176 species) tick genus.5 This eyeless genus can, in most cases, be identified by a pronounced lateral projection of palpal segment 2, which extends well beyond the basis capitulum. In Europe, TBEV has been recovered from Haemaphysalis punctata (the red sheep tick), Haemaphysalis concinna in Europe and Asia, and from Haemaphysalis longicornis in Asia.9
The genus Hyalomma is relatively small with 27 species of small- to large-sized ticks.14 They are characterized by their elongated palps, which are at least twice as long as wide. The distinct eyes are located in sockets adjacent to the postero-lateral edges of the scutum that is unornamented. The distribution of Hyalomma species is limited to the Old World, primarily to arid or semiarid habitats. Hyalomma marginatum (the Mediterranean Hyalomma) is the only member of this genus from which TBEV has been recovered.15,16
The biology of hard ticks
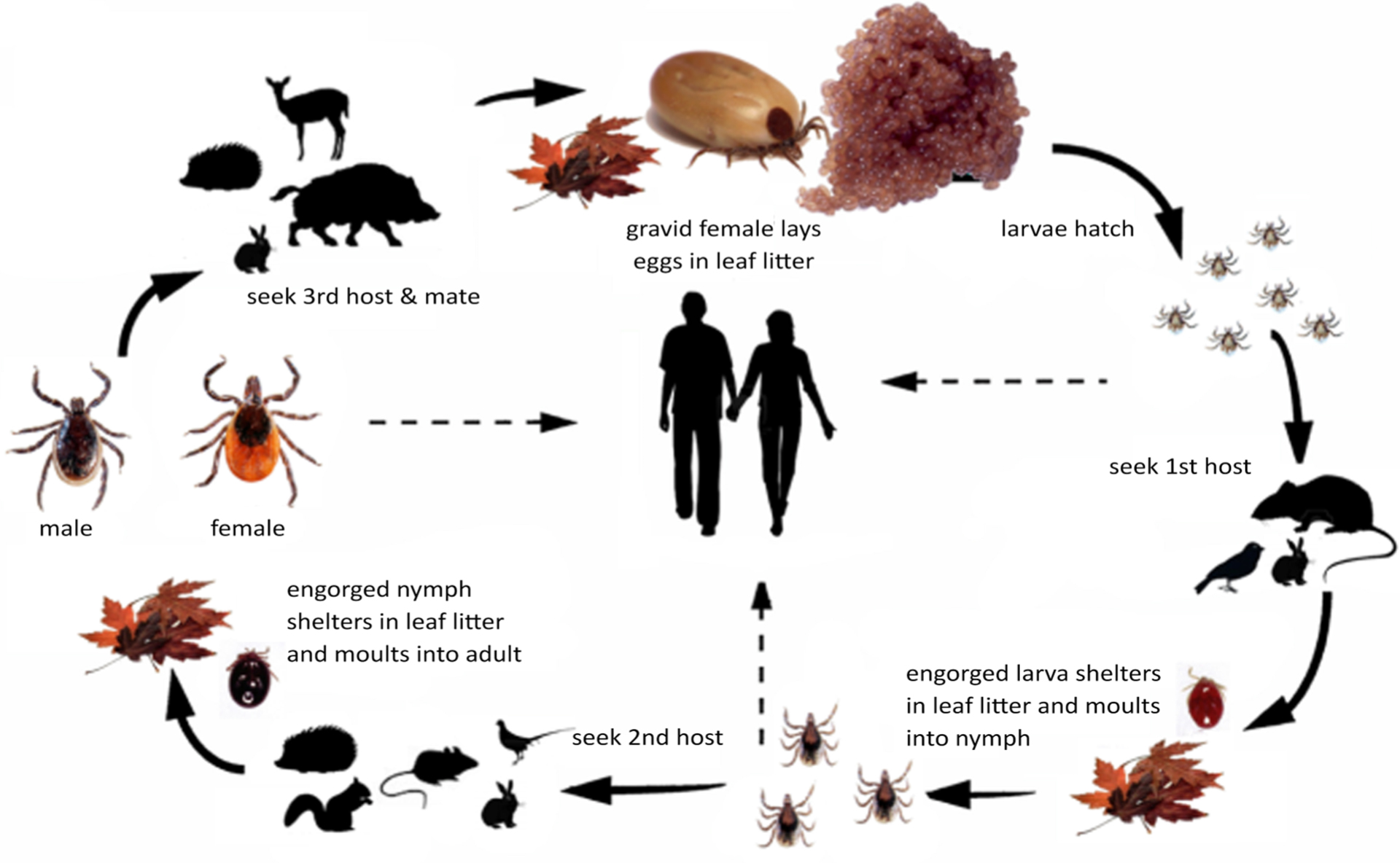
All the species known to transmit TBEV have a 3-host life- cycle (Figure 2). Each postembryonic life stage requires a blood meal from a suitable host, after which the tick detaches and molts in the leaf litter. The arrows with broken lines in the figure show the potential transmission paths to humans. The line from larvae to humans indicates that transovarial trans-mission from an infected female can happen which results in infective larvae. Infection of the tick can occur when larvae, nymphs, or females feed on an infective host (see below).
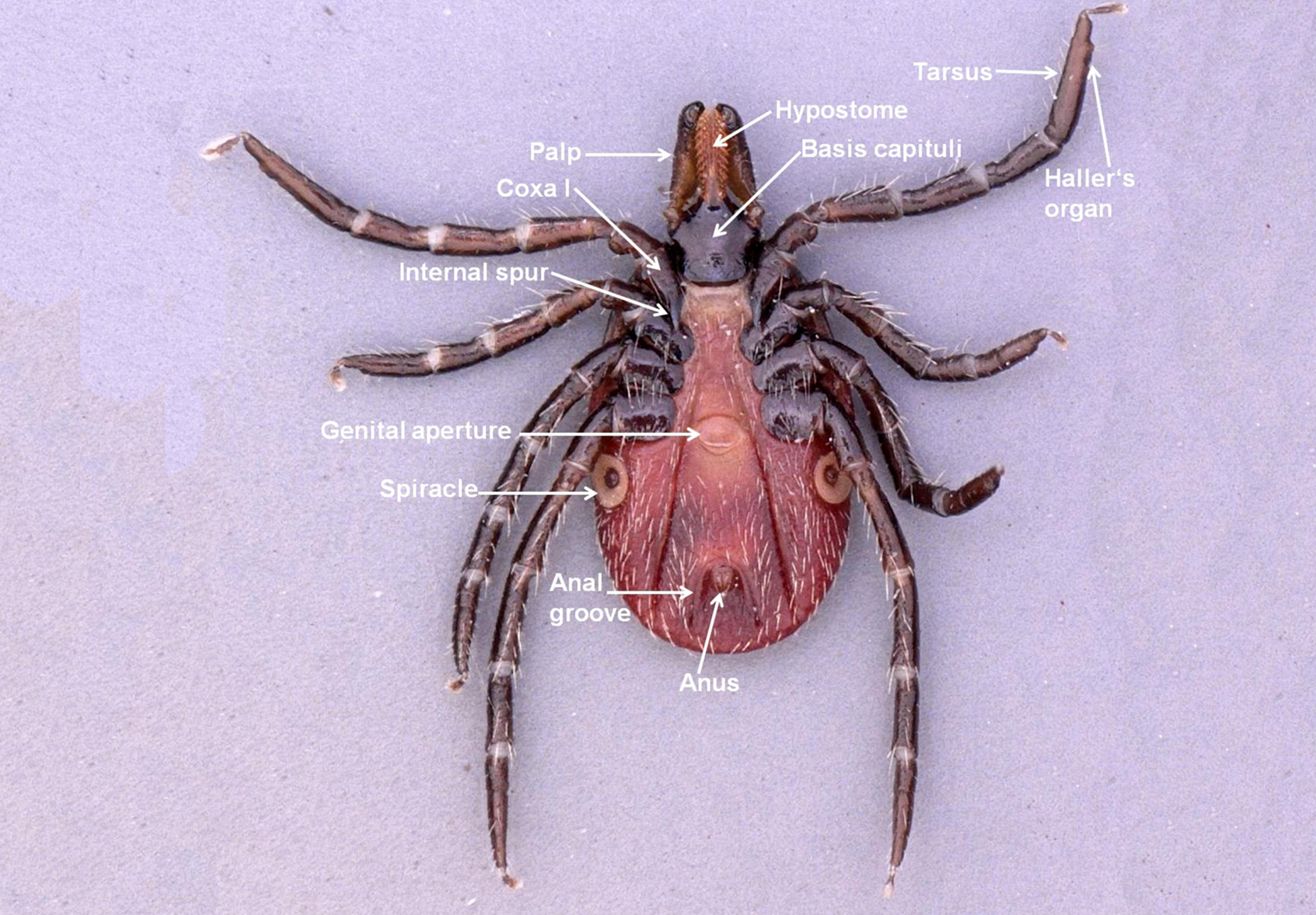
The larva, nymph, and adult (female or male – Figures 3a, 3b, 3c, and 3d) are active stages that require a host (this is not the case for males of the genus Ixodes, which can mate off-host without feeding).17 Larvae are easily recognizable by the presence of only 3 pairs of legs, and absent spiracular and genital apertures (Figures 4a and 4b). Nymphs have 4 pairs of legs and spiracles (Figures 5a and 5b). Adult females have 4 pairs of legs, and spiracles, a genital aperture, and porose areas on the dorsal surface of the basis capituli (Figures 3a and 3b). Adult males have 4 pairs of legs, the scutum covers the entire dorsal surface, and seven hard sclerotized plates cover the ventral body surface of some species (Figures 3c and 3d).
Table 1: Tick species, habitats, and involved hosts in relation to the TBEV subtype and distribution
ES, European subtype (TBEV-EU); FES, Far Eastern subtype (TBEV-FE); SS, Siberian subtype (TBEV-Sib)
* Reference for tick habitat and host: Nowak-Chmura and Siuda, 2012;36Petney et al., 2012;17
Guglielmone et al., 201412
** References for tick species involved in TBE virus transmission
Types of hard ticks
Ixodid ticks fall into two behavioral groups. Exophilic or non-nidicolous ixodid ticks occur in the open environment and are associated, with forests, savannahs, second-growth areas of scrub and brush, grassy meadows, semi-desert, or desert areas. These species are usually not very host-specific. Nidicolous or endophilic ixodid ticks live in or near the nests of their hosts, are adapted to highly specialized environments (crevices or other shelters used by their hosts), and tend to be more host-specific.9 Many Ixodes species are nidicolous.9 The main vectors of TBEV, I. ricinus and I. persulcatus are exophilic and exceptional both in terms of their large variety of hosts they use as well as the habitats they occupy.37
Figure 2: The life-cycle of Ixodes ricinus
The dotted arrows indicate potential transmission to humans.©Nina Littwin
Click the image above to enlarge
Click the image above to enlarge
Click the image above to enlarge
Click the image above to enlarge
Click the image above to enlarge
Host-finding behavior
Ixodid ticks’ host-seeking behavior is under the control of different abiotic factors that differ according to the region. In temperate and sub-polar regions, seasonal activity is mainly regulated by ambient temperature, changing photoperiod, and incident solar energy, and in the more temperate regions, tick activity is often controlled by saturation deficit and relative humidity, with long-term dry conditions being adverse for survival.38-41 Those species involved in the transmission of TBEV tend to quest passively or ambush their hosts by climbing onto weeds, grasses, or other lower vegetation to wait for a host to pass nearby.
Ixodes ricinus adults can climb as high as 1.5 m on brushy vegetation. The immature stages are found lower, up to 70 cm for larvae (O. Kahl, personal communication) and less than 1 m for nymphs.9 Ticks are able to sense a host with their Haller’s organ, which is located on the tarsus I. Haller’s organ possesses chemo-, mechano-, and thermoreceptors that also ensures (together with the receptors on the palps) selection of a suitable feeding site on the host body. The most important stimuli are carbon dioxide (CO2), vibration produced by moving potential hosts, and host temperature. For some species, visual images, host smell, and even noise can stimulate the tick.9,42,43
Feeding behavior
Feeding behavior, even on preferred hosts, is not a uniform process. An ixodid tick may crawl on the host for several hours in search of a suitable feeding site. After attachment, many ixodid ticks secrete cement during the first 1–2 days to secure themselves at the wound site.42 The feeding tick begins salivating into the developing hematoma and sucking blood; phases of salivation and blood sucking alternate.9 Saliva not only plays an important role in the feeding tick’s osmoregulation44 but also has a variety of pharmacological effects. There is an extensive array of antihemostatic, anti-inflammatory, and immunomodulatory proteins and lipids in the tick saliva that suppress the host’s ability to reject the feeding tick.9,45-47 Anticoagulant effects, inhibiting factor Xa, were first shown in I. ricinus in 1898-1899.48 In addition, many tick species produce proteins that inhibit thrombin directly or inhibit the conversion of prothrombin to thrombin by inhibiting factor V. Other proteins prevent platelet aggregation or bind, antagonize or degrade important host mediators of pain, itching and inflammation, particularly the host’s own histamine, serotonin, and bradykinin.9,47
Ixodid ticks feed gradually because they must first produce new cuticle to accommodate the massive blood meal.9 Typical attachment periods range from as few as 2 days for larvae to as long as 13 days for females.49
An I. ricinus female can reach approximately 450 mg at the end of feeding from approximately 2 mg at the beginning of feeding.50
Drop-off
The controlled timing of drop-off from the host offers important ecological advantages. For non-nidicolous ticks, such drop-off rhythms are synchronized with host behavioral patterns. This tends to disperse fed ticks in optimal habitats where they can develop and reproduce. Photoperiod appears to be the dominant abiotic exogenous factor affecting drop-off patterns. The daily light:dark cycle induces a regular rhythm of feeding and dropping off. Detachment may occur while hosts are inactive in their nests or burrows or, alternatively, it may be coordinated with the period of high host activity.50
Host specificity
Tick species can be either opportunistic or specific with respect to the hosts they choose; both I. ricinus and I. persulcatus are opportunistic species, especially the immatures. For I. ricinus, more than 300 species of vertebrate hosts have been recorded.9,28 Larvae and nymphs of I. ricinus feed readily on lizards, birds, and small mammals, as well as on larger hosts including deer. Adults feed on medium-sized and large mammals, especially ungulates, as well as humans, as do the immature ticks.9 Ixodes persulcatus is more restricted to 46 species of hosts.51,52
Questing height is also important. Ticks questing on or near the ground are exposed mostly to small animals, while those questing higher in the vegetation are more likely to encounter larger animals. The extent to which different hosts are utilized depends on host behavior and opportunities for contact, such as foraging range, time of day and time spent foraging, habitats visited, and other factors.14
Acceptance of a vertebrate animal is also dependent on physiological factors and the ability of the ticks to recognize it as a host. Host utilization may be influenced by the ability of ticks to evade or suppress host homeostatic systems and avoid rejection.47
Hard tick ecology, environmental factors
Ticks occur in many terrestrial habitats ranging from cool, arboreal northern forests to hot, arid deserts. Each species, however, has become adapted to the specific types of habitat where it is generally found in highest abundance. All I. ricinus postembryonic stages are exophilic and depend entirely on a suitable combination of climatic variables, making them vulnerable to climate changes and especially to desiccation. Thus, they are mainly found in cool, moist forests.42,53,54
Water balance is a critical determinant of a tick’s ability to wait for hosts. Ticks may quest for weeks or even months while waiting for a host. When they have a body water deficit, they retreat to more sheltered, humid micro-environments, such as the rotting vegetation in a meadow or damp leaf litter on the forest floor. They secrete a hygroscopic salivary secretion onto their external mouthparts that collects atmospheric water at relative humidity = 80-85% (active water vapor sorption).42 Rehydrated ticks are able to resume host-seeking. Some ticks are able to remain in the questing position for many days without rehydration, while others must return to their humid microenvironments.55 Dense ecotonal vegetation provides shade, increased moisture, protection from intense solar radiation, and plants that support the tick hosts.
There have been various studies showing the relationship between I. ricinus and vegetation type in central Europe56,57 and the capacity of this species to adapt to a large variety of biotopes with low temperature (e.g., Sweden) and high altitudes, up to 1500 m.58-60
Ixodes persulcatus is distributed in 14 countries, between 21° and 66° of northern latitude in Eurasia, mostly with a temperate continental climate.51 In a model predicting the suitable habitats for I. persulcatus, it was shown that temperature and humidity are the main factors in the distribution of this species.51 Vegetation also has an impact on the tick distribution, its requirement is wood and wet biotopes.51
Normally, temperature and relative humidity in a burrow, cave, or similar type of shelter are more uniform throughout the year than in the external macro-environment. The higher relative humidity in such microenvironments is due in part to the presence of hosts, their wastes, and the plant materials they use to construct or line their nests.61 Nidicolous ticks exhibit behavioral patterns that restrict their distribution to these sheltered locations. They avoid bright sunlight and low humidity, the type of conditions prevailing at the entrances of burrows or caves. Confined within these hidden, restricted locations, nidicolous ticks become active when hosts are present. However, when the hosts are absent, they may wait for up to several years for hosts to return, or until they die of starvation.
Diapause
An important physiological trait that enables ticks to survive adverse environmental conditions and conserve energy until conditions improve is diapause as a form of dormancy.62 Diapause is induced by an external cue before adverse conditions occur. It is not terminated by favorable external conditions – as it is the case with quiescence – but there is some diapause development before its termination. During diapause ticks become inactive, reduce their metabolic rates, and do not feed on hosts even when given the opportunity.53 Diapause can occur in each life stage, whether it is unfed or engorged. This varies, however, between species and can also differ within a tick species in different geographic areas. As an example, oviposition can be delayed in D. marginatus. Engorged females that feed in late summer, early fall or in winter oviposit only in the following spring.53
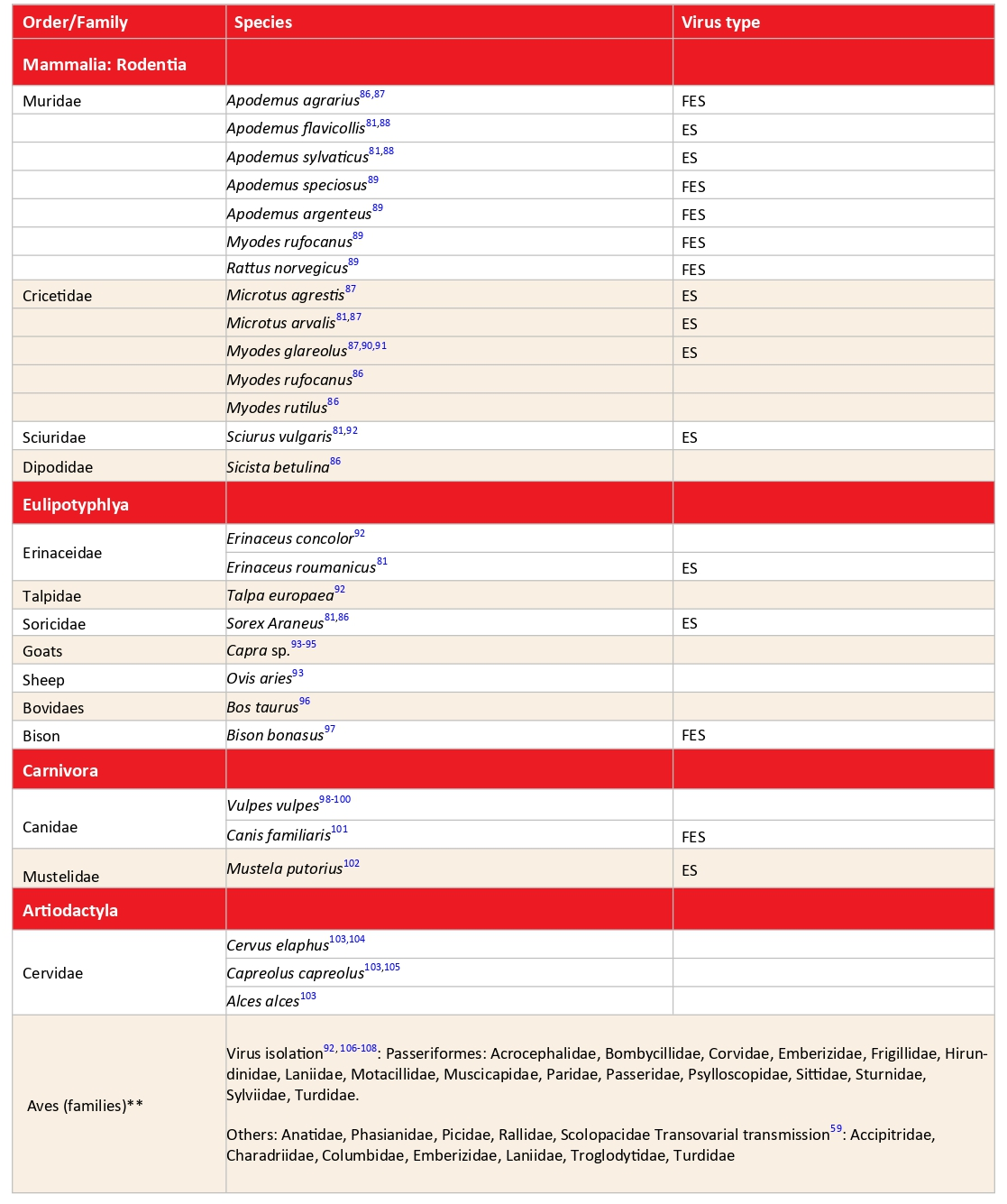
Table 2: Animal hosts from which TBEV* has been recovered
ES, European subtype (TBEV-EU); FES, Far-Eastern subtype (TBEV-FE); SS, Siberian subtype (TBEV-Sib)
*Selected references; **Less information available
Life cycle and seasonal activity
Ixodes persulcatus inhabits mainly coniferous forests of Asia and Eastern Europe, while I. ricinus inhabits deciduous and mixed forests in the British Isles, in Continental Europe, and western Asia.51,53,63 Ixodes persulcatus adult females and eggs are unable to survive the winter, however, that I. persulcatus larvae and nymphs, whether unfed or engorged, are able to overwinter. In contrast, eggs as well as unfed and satiated females of I. ricinus are capable of overwintering, a principal difference between the life-cycles of the two tick species. Vector tick activity is well correlated with the seasonal pattern of TBE occurrence. In such a focus, it is common for 2–3% of the ticks to be virus-infected.63 In Northern and Central Europe, the seasonal activity of I. ricinus often has 2 peaks, one in spring (May–June) and the other one at the end of summer (September-October).
For I. persulcatus adults four types of seasonal dynamics throughout their distribution area were described, differing in the duration of the active period.63,64 In the north-western area of distribution, I. persulcatus becomes active immediately after the melting of the snow cover with a rapid increase in abundance in May, followed by a sharp decline in mid-summer. In Karelia (a middle taiga subzone), adult activity lasted on average 74 days. Between 2012 and 2023, the relative abundance of ticks increased significantly in comparison with the 1980s monitoring period, showing a tendency towards an earlier start of the tick activity, as in the 1980s.63
Unfed Dermacentor reticulatus adults are mostly active in spring and autumn, occasionally in winter, but usually not in summer (June to early August).65-67 During periods of snow cover and the driest and hottest weeks of the year Dermacentor reticulatus is inactive.68-69 The larvae feed for 3-6 days, nymphs for 5-12 days, and females for 7-16 days, while males may remain in the host for a long time, even in the absence of females.51-52 Adults can overwinter unfed or engorged53,54 and are able to survive 2.5 years of starvation.55 Interestingly, this tick can spend the whole winter on hosts.49 Dermacentor reticulatus eggs can survive under water for several months and may be spread by floods into new areas.66 Dermacentor reticulatus in immature life stages is assumed to be nidicolous and therefore cannot be collected from vegetation. Nevertheless, Schmuck et al.70 collected D. reticulatus immatures (47 questing larvae and two nymphs) by flagging in June and July in 2018 and 2019, in two different locations close to the city of Leipzig, Germany. To understand under which circumstances D. reticulatus immatures were found outside the burrows of their hosts and can be collected from vegetation needs further investigation.70
Tick species involved in TBEV transmission
Of the 54 species of ixodid ticks known from the Western Palearctic,51 eight species from three genera are known to be able to transmit TBEV, and the virus has been isolated from at least 14 other species (Table 1). Ixodes ricinus, the most commonly encountered European tick species, is considered to be the principal vector of TBEV there.71 Lichard and Kožuch27 were able to show TBEV persistence and transmission to white mice by Ixodes arboricola, which is considered a secondary amplifying vector of TBEV.72 Ixodes persulcatus is also known to transmit TBEV.73-75 It is the adult female I. persulcatus, which infects humans with TBEV and other zoonotic pathogens. Neither the larval nor the nymphal stage often attaches to humans.9 Both D. marginatus and D. reticulatus are also vectors of TBEV.24,65,76,77
Haemaphysalis concinna is a known vector of TBEV as well.78,79 Evidence for the vectorial capacity of Haemaphysalis inermis for TBEV is available from Nosek et al.80 The virus has been isolated in the Czech Republic from female and nymphal I. hexagonus infesting a hedgehog.29,81,82 TBEV also has been detected in Haemaphysalis punctata.83
The role of Dermacentor ticks (Table 1) in the circulation of TBEV in the environment is unclear and poorly studied.65 D. reticulatus appears to be spreading and population density increasing during recent decades.66,84,85 In eastern Poland, the mean prevalence of infection with TBEV found in questing adult D. reticulatus was 10.8% (range 7.3–14.3% in infected areas): This is considerably higher than the prevalence found in questing adult I. ricinus (1.6%, range 0.7–4.3% in infected areas).21,
Prevalence of TBEV in questing adult D. reticulatus ticks from Białowieża Primeval Forest was similar (1.58%)24 to that in questing I. ricinus (1.30%),77 as was the case in Moldova (adult I. ricinus 3.8%, adult D. reticulatus 3.9%, but adult Haemaphysalis punctata 8.8%).80 The natural occurrence of TBEV in a D. reticulatus tick population was also proven for Germany during 2016 to 2018 by isolation of several TBEV strains from this tick species in a natural focus.65
The differences in TBEV prevalence in the various vector species remain puzzling. Questing I. ricinus usually have a very low prevalence of the virus, ranging from no virus in many areas to less than 1% in most others, and rarely reaching 2–5%, in unfed adults.111-114 Knap and Avsic-Zupanc115 showed that over a 4-year period, the prevalence was at the expected low level in the 8 areas studied, but that no area was consistently positive for the virus. This may be related to the frequently low sample sizes (14/30 samples had fewer than 300 specimens).
Prevalence of the virus in feeding ticks, although very variable, can be substantially higher.116 Waldenström et al.117 showed a low prevalence (0.5%) in nymphs and larvae feeding on migratory birds in Sweden, while Kazarina et al.106 detected 14% nymphs and 7% larvae of I. ricinus on migratory birds infected in Latvia. Data for I. persulcatus are more variable. Korenberg and Kovalevskii118 reported a high TBEV prevalence in unfed adults, ranging from 10.9% to 38.7% over 6 years (mean 26.2%) in unfed adults in the Pre- Chapter 5: TBEV-transmission and natural cycles Ural Region, whereas the prevalence in the Primorskii Region of the Russian Far-East ranged from a little over 1% to over 9% from 1970 to 1990, and in the Khabarovsk Region from 3.4% to 9.4% over 4 years.119 In the Novosibirsk Region, the prevalence of TBEV in unfed adult I. persulcatus was 3.6%, with 0.8% being pathogenic to laboratory mice.86 In the same study, 3.3% of questing adult I. pavlovskyi were infected with the virus with 1.8% of the isolates being pathogenic. Information on less commonly encountered vectors is rarely available and sample sizes are usually low, making such data unreliable (e.g., Kim et al.)120 Long-term studies and statistical analyses showed that higher average temperatures during the summer-autumn period may lead to higher levels of TBEV found in ticks and consequently increase the risk for humans to develop symptomatic TBE following an infected tick bite.121
Vertebrate hosts
The prevalence of antibodies to TBEV in hosts is quite variable.122 TBEV has been found in numerous mammal species from different families, as well as in a large number of passerine and non-passerine bird species (Table 2). Virus infection was demonstrated by antibodies to the virus or viral ribonucleic acid (RNA) detection in a wide variety of bird species,117,123 with virus isolation from Turdus pilaris (fieldfare) and Acrocephalus dumetorum (Blyth’s reed warbler) opening the possibility of virus transfer to new foci during bird dispersal or migration. Viremia has been induced experimentally in birds, reaching levels sufficient to infect feeding ticks.123,124 Generally speaking, findings of TBEV in animals, whether indirect or direct, do not mean very much eco-epidemiologically. Only the demonstration of reservoir competence indicates an active role in the perpetuation of TBEV.
Red foxes (Vulpes vulpes) are known to be reservoir-competent for TBEV.99,125 Although I. hexagonus is a proven vector of TBEV, little is known about the vector competence of the fox tick I. canisuga.
In recent years, the detection of viral RNA in hosts has become possible. Tonteri et al.,126 in Finland, detected the European (TBEV-EU) and Siberian (TBEV-Sib) subtypes in M. glareolus, TBEV-Sib in the shrew Sorex araneus, and TBEV-EU in Microtus agrestis. Achazi et al.87 detected TBEV RNA in rodent brain tissue in prevalences up to 20% in TBE non-risk as well as in risk areas in east-German Federal States. In the Novosibirsk region of Siberia, where I. persulcatus and I. pavlovskyi are the main TBEV vectors, the prevalence of TBEV viral RNA in five small mammal species was extremely high.86 It ranged from 35.3% for A. agrarius organs to 82.2%for Myodes rutilus blood, with a mean value for all species and tissues of 62.1%. All 3 virus subtypes were represented. In addition to small mammal hosts, larger wild and domestic animals frequently have high antibody prevalences. Because they feed large numbers of vector ticks, they can be used as sentinels for the occurrence of TBEV in a given area.
TBEV transmission
Nuttall et al.127 noted: “Reciprocal interactions of parasites transmitted by blood-sucking arthropod vectors have been studied primarily at the parasite-host and parasite-vector interface. The third component of this parasite triangle, the vector-host interface, has been largely ignored.”
The adult female tick is considered to play only a minor role in virus circulation. Tick males, which either do not feed or feed for only a short time, might also be involved in virus transmission.128 TBEV invades all tick tissues, including the salivary glands and ovaries,16 thus it may be transmitted by ticks in the following ways: 1) via saliva, 2) transovarially (vertically), and 3) sexually.128-130
TBEV transmission from vector ticks to hosts via saliva
Certain species of ticks are vectors and reservoirs of TBEV, and they can transmit the virus already when they start feeding131-132 with viral particles contained in the saliva, which the ticks release into the host tissues.133 Tick-borne virus transmission occurs within 24 h of tick attachment, possibly within the first hour of feeding (Ebel et al. 2004). After attachment, many ixodid ticks secrete cement to fix themselves in the skin, but in a study, Alekseev et al. (1996) showed that cement plug may act as bolus of infection for TBEV and Borrelia burgdorferi s.l. When an infected tick had been removed, the remaining cement cone embedded in the skin resulted in TBEV infection within one hour of detachment.18
TBEV is present in the alveolar cells of the salivary glands of D. marginatus and H. inermis females in as few as five days after their feeding on viremic white mice.135 Also certain vertebrates, so-called reservoir hosts, are important for the amplification of the virus and are together with vector ticks the basis for the heteroxenous TBEV perpetuation.136
Viremic transmission from hosts to feeding ticks
Ticks become infected with TBEV while they feed on a viremic host.63,137 Nosek et al.138,139 proved that a viremia in a host lower than 101 mouse LD50./0.03 ml was insufficient to cause infection in ticks. In individual engorged I. ricinus ticks, the virus titer was 101-104 mouse LD50/0.03 ml. Viremic white mice served as virus donors. Grešíková and Nosek80 demonstrated the persistence of TBEV in H. inermis (from larva to nymph) and then the transmission from H. inermis nymphs to white mice. Viremia surpassing the threshold values of infectivity for tick vectors was also found in some juvenile and adult Myodes rufocanus, M. rutilus, and Micromys minutus. The viremia level depends on the rodent species and age and exhibits individual variability.140
Co-feeding transmission
TBEV transmission is also possible from infected to non-infected ticks during feeding close to each other on a non-viremic host.141-142 Cellular infiltration of tick feeding sites, and the migration of cells from such sites, can provide a vehicle for transmission between co-feeding ticks that is independent of host viremia.143 The non-viremic route of transmission between co-feeding ticks can even occur in rodents that are already immune to TBEV.144 The degree of co-feeding virus transmission may be influenced by local climatic factors that affect the seasonal timing of tick host-seeking activity and, as such, can be used to predict the focal distribution of TBEV.141-142
Transovarial transmission
Another possible way for ticks to transmit TBEV involves transovarial transmission and transstadial persistence (see below), which were described for the first time as early as 1940.145 However, only some eggs in the batch of a TBEV-infected vector tick female become infected.130 In addition, virus can partly be lost during transition from stage to stage,146 and not all tick individuals reach the next life stage irrespective of the presence or absence of the pathogen. Danielova and Holubova130 found that only 0.23% of larvae coming from infected females were TBEV-positive. Other studies showed that 0.58% to 0.75% of the larvae were transovarially infected. Thus, the rate of transovarial transmission remains below 1%. Nuttall et al.147 suggest that transovarial transmission is important for the maintenance of a natural focus even if it occurs at a very low rate.
Danielova et al.148 detected TBEV in 2 out of 647 flagged larvae of I. ricinus, which indicates transovarial transmission.
Transstadial persistence
TBEV was not detectable in I. ricinus nymphs 14 days after molting from larvae that had engorged on viremic A. flavicollis, but TBEV was present in these ticks two months post ecdysis. Many nymphs contained the virus, indicating that the latter undergoes an eclipse phase during metamorphosis.149
Sexual transmission in ticks
Transmission of TBEV from males to females128 is successful in only 10% of copulations in infected I. persulcatus, but it may provide notable support for the transfer of the virus to the following generation of ticks if transovarial transmission follows. A mathematical model of sexual transmission of the virus150 was developed long before determining that such a sort of transmission occurs. Virus exchange between a non-engorged female and an infected male of I. persulcatus that ‘feeds’ on (i.e., attaches to) the female before or after copulation is quite probable, and it has been proven that the saliva of starved males contains a fairly large amount of virus, sufficient for infecting not only animals but also humans. The feeding of I. persulcatus males on females with which they later copulate can be observed in 2–10% of cases.151,152
Vertical TBEV transmission in vertebrates
TBEV transmission from mother to her offspring in small rodents, e.g., red voles (M. rutilus), was shown for naturally infected reservoir hosts as well as after experimental infection with different sublethal doses of the virus.153 TBEV RNA was detected in up to 90% of the newborn rodents, 240–280 days after experimental infection of their parents, by real-time polymerase chain reaction (RT-PCR), enzyme-linked immunosorbent assay (ELISA), and bioassays. The small amounts of TBEV RNA detected in the embryos, placenta, and blood serve as evidence of prenatal transmission. Postnatal transfer of the virus might occur through the rodent’s milk. Vertical virus transmission may occur before, during, and/or after birth of the baby rodents with a high frequency. In natural foci, this could ensure long-term persistence of TBEV in mammal hosts149 without involving any arthropod vectors.86 Divé et al.154 reported detailed investigation of pre-and postnatal health assessment of three children in the context of severe maternal TBEV infection during pregnancy. The clinical and virological data strongly suggest that fetal TBEV infection did not occur, despite severe manifestations in the pregnant females. Non-reservoir hosts do not directly participate in virus transmission but can play an important role in the maintenance of natural foci. The density of reservoir-incompetent hosts may have either a positive effect on virus transmission, by amplifying the tick population, or a negative (‘dilution’) effect, as tick bites on a non-reservoir host cannot lead to virus transmission.155,156
Alimentary route of transmission
Humans mostly become infected with TBEV via tick bites, but viral transmission is also possible via the consumption of unpasteurized goat, cow and sheep milk.157 Approximately 1% of all TBEV infections in humans are probably acquired by consuming infected unpasteurized milk and milk products from infected livestock, particularly goats.158
Outbreaks due to alimentary virus transmission are known from Eastern, Central and Southern Europe, and have to be considered particularly in cases of local epidemics.162-164 Ličková et al.165 summarize the history and recent alimentary TBEV infections in Europe. In an alimentary outbreak in Germany, due to consumption of a fresh goat cheese, the virus could be for the first time isolated from naturally infected cheese.164
TBEV interhuman transmission of TBEV by breast milk has not been confirmed or ruled out. Kerlik et al.166 reported a case of probable transmission of TBEV from an unvaccinated mother to an infant through breast-feeding.
The natural cycle
The natural cycle of TBEV is highly complex, and many details remain obscure. The three prevailing TBEV subtypes overlap in some areas, they all have multiple mammalian reservoir hosts and various tick vectors, and in some areas these subtypes occur sympatrically. Humans are not included in these natural cycles, but may enter those trans-mission cycles inadvertently.
Small mammals as a reservoir and vector ticks play a central role in the natural cycle of TBEV, but non-reservoir hosts such as birds and large vertebrates, such as wild ungulate species, or foxes, may also indirectly contribute to the spread and maintenance of TBEV. Additionally, changing climatic patterns, as well as changes in ecosystems, may not only affect the spatial distribution of TBEV, but also the maintenance of small natural TBEV foci.167,168 Small rodents such as A. flavicollis are important hosts for the larvae of I. ricinus, the probably most important TBEV amplifying host in Central Europe. Apodemus flavicollis temporarily develops high virus titers necessary to infect ticks. Detailed studies by Radda et al.,98,149 who trapped small rodents and collected the engorged ticks in a natural TBE focus for 2 years, showed that given certain prerequisites are fulfilled (high numbers of rodents, vector tick larvae and nymphs feeding on these rodents), such a natural TBEV focus is able to sustain itself without any significant input of other hosts. This may explain why many of these natural foci are stable, but restricted to small areas, and why they harbor TBEV-positive ticks over a long period of time. Forest structure, especially deforestation and reforestation, are known to have a huge impact on ticks and vertebrate reservoir hosts for many tick-borne pathogens.159,160
Experimental transstadial maintenance of TBEV in D. marginatus and D. reticulatus ticks emphasizes the role of both species. TBEV infection and transmission rates in Dermacentor species to hosts are somewhat lower than in species of the genera Ixodes and Haemaphysalis.161 Feeding larvae and nymphs of I. persulcatus may become infected with TBEV if the virus titer in the host blood reaches at least 3.0 log10 LD50/0.03 mL.151 Such levels of viremia occur only in small rodents and are a critical factor in the virus circulation between vertebrates and ticks in natural foci. In small rodents, the infection is asymptomatic.90
TBEV has been isolated from a wide range of birds from many different families, including migratory species, which may be important for the distribution of the virus. A common strategy for migratory birds is to rest at certain stopover sites along their routes. At these sites, the birds can be infested with ticks or engorged ticks can detach after engorgement. Sándor et al.169 detected 4 different tick species on 11 different bird species in the Danube Delta, including larvae, nymphs, and females of I. ricinus.
A high variability is found between areas and years with respect to viral prevalence in both vertebrate hosts and vector tick populations, while consistent differences between vectors. For example the generally higher TBEV prevalences in I. persulcatus compared with those in I. ricinus may relate to the ecology/biology of the individual vectors. The complexity is well defined by the various mathematical models aimed at exploring the dynamics of TBEV ecology156,170,171 Hartemink et al.171 list 19 parameters based on field data to define the basic reproduction number (Ro) of tick-borne infections, while Rosà et al.156 list 32 parameters in a more comprehensive model. Unfortunately, no single study has been able to comprehensively measure all the parameters needed to test these models, although approximations are available.
Contact
Lidia Chitimia-Dobler
lydiachitimia@gmail.com
Author
Citation
Chitimia-Dobler L. TBEV-transmission and natural cycles. Chapter 5. In: Dobler G, Erber W, Bröker M, Chitimia-Dobler L, Schmitt HJ, eds. The TBE Book. 7th ed. Singapore: Global Health Press; 2024.doi:10.33442/26613980_5 5-7
References
- de la Fuente J, Estrada-Pena A, Venzal JM, Kocan KM, Sonenshine DE. Overview: Ticks as vectors of pathogens that cause disease in humans and animals. Front Biosci. 2008;13:6938-6946. doi:10.2741/3200.
- Estrada-Peña A, Jongejan F. Ticks feeding on humans: a review of records on human-biting Ixodoidea with special reference to pathogen transmission. Exp Appl Acarol. 1999;23(9):685-715. doi:10.1023/a:1006241108739.
- Skuballa J, et al. Molecular detection of Anaplasma phagocytophilum in the European hedgehog (Erinaceus europaeus) and its ticks. Vector Borne Zoonotic Dis. 2010;10(10):1055-1057.
- Pfäffle M, et al. The ecology of tick-borne diseases. Int J Parasitol. 2013;43(12-13):1059-1077.
- Guglielmone AA, Nava S, Robbins RG. Geographic distribution of the hard ticks (Acari: Ixodida: Ixodidae) of the world by countries and territories. Zootaxa. 2023;5251(1):1-274.
- Estrada-Pena A, Guglielmone AA, Nava S. Worldwide host associations of the tick genus Ixodes suggest relationships based on environmental sharing rather than on co-phylogenetic events. Parasit Vectors. 2023;16(1):75.
- Barker SC, Burger TD. Two new genera of hard ticks, Robertsicus n. gen. and Archaeocroton n. gen., and the solution to the mystery of Hoogstraal’s and Kaufman’s “primitive” tick from the Carpathian Mountains. Zootaxa. 2018;4500(4):543-552.
- Mans BJ, et al. Nuclear (18S-28S rRNA) and mitochondrial genome markers of Carios (Carios) vespertilionis (Argasidae) support Carios Latreille, 1796 as a lineage embedded in the Ornithodorinae: re-classification of the Carios sensu Klompen and Oliver (1993) clade into its respective subgenera. Ticks Tick Borne Dis. 2021;12(4):101688.
- Mullen GR, Durden LA. Medical and veterinary entomology. 3rd ed. London, United Kingdom: Academic Press, an imprint of Elsevier; 2019.
- Petney TN, et al. A Look at the World of Ticks. In: Progress in Parasitology. 2011:283-296.
- Sonenshine DE, Roe RM. Biology of ticks. 2nd ed. New York: Oxford University Press; 2014
- Guglielmone AA, Nava S. Names for Ixodidae (Acari: Ixodoidea): valid, synonyms, incertae sedis, nomina dubia, nomina nuda, lapsus, incorrect and suppressed names–with notes on confusions and misidentifications. Zootaxa. 2014;3767:1-256.
- Clifford CM, et al. Systematics of the Subfamily Ixodinae (Acarina: Ixodidae). 1. The Subgenera of Ixodes. Ann Entomol Soc Am. 1973;66(3):489-500.
- Sands AF, et al. Effects of tectonics and large-scale climatic changes on the evolutionary history of Hyalomma ticks. Mol Phylogenet Evol. 2017;114:153-165.
- Yurchenko OO, et al. Partial Characterization of Tick-Borne Encephalitis Virus Isolates from Ticks of Southern Ukraine. Vector Borne Zoonotic Dis. 2017;17(8):550-557.
- Nawrocka E. Studies on the experimental infection of Hyalomma dromedarii (Koch) ticks with tick-born encephalitis virus. Acta Microbiol Pol. 1978;27(1):19-24.
- Petney TN, Pfäffle MP, Skuballa JD. An annotated checklist of the ticks (Acari: Ixodida) of Germany. Syst Appl Acarol. 2012;17(2).
- Alekseev AN, et al. Preliminary studies on virus and spirochete accumulation in the cement plug of ixodid ticks. Exp Appl Acarol. 1996;20(12):713-723.
- Demina TV, et al. Genotyping and characterization of the geographical distribution of tick-borne encephalitis virus variants with a set of molecular probes. J Med Virol. 2010;82(6):965-976.
- Süss J. Tick-borne encephalitis 2010: epidemiology, risk areas, and virus strains in Europe and Asia—an overview. Ticks Tick Borne Dis. 2011;2(1):2-15.
- Wojcik-Fatla A, et al. Prevalence of tick-borne encephalitis virus in Ixodes ricinus and Dermacentor reticulatus ticks collected from the Lublin region (eastern Poland). Ticks Tick Borne Dis. 2011;2(1):16-19.
- Stefanoff P, et al. Virus detection in questing ticks is not a sensitive indicator for risk assessment of tick-borne encephalitis in humans. Zoonoses Public Health. 2013;60(3):215-226.
- Katargina O, et al. Detection and characterization of tick-borne encephalitis virus in Baltic countries and eastern Poland. PLoS One. 2013;8(5).
- Biernat B, et al. Prevalence of tick-borne encephalitis virus (TBEV) RNA in Dermacentor reticulatus ticks from natural and urban environment, Poland. Exp Appl Acarol. 2014;64(4):543-551.
- Drelich A, et al. Prevalence of tick-borne encephalitis virus in a highly urbanized and low risk area in Southern Poland. Ticks Tick Borne Dis. 2014;5(6):663-667.
- Cuber P, et al. Risk of exposure to ticks (Ixodidae) and the prevalence of tick-borne encephalitis virus (TBEV) in ticks in Southern Poland. Ticks Tick Borne Dis. 2015;6(3):356-363.
- Lichard M, Kozuch O. Persistence of tick-borne encephalitis virus in nymphs and adults of Ixodes arboricola and its transmission to white mice. Acta Virol. 1967;11(5):480.
- Gresikova M, Kaluzova M. Biology of tick-borne encephalitis virus. Acta Virol.1997;41(2):115-124.
- Krivanec K, et al. Isolation of TBE virus from the tick Ixodes hexagonus. Folia Parasitol (Praha). 1988;35(3):273-276.
- Valarcher JF, et al. Tick-borne encephalitis. Rev Sci Tech. 2015;34(2):453-466.
- Radda A, Pretzmann G, Steiner HM. Field investigations on the ecology of tick-borne encephalitis (TBE-CEE) virus in lower Austria. J Hyg Epidemiol Microbiol Immunol. 1968;12(3):274-283.
- Streissle G. [Studies on the transmission of the virus of early summer meningoencephalitis by the tick Ixodes hexagonus Leach]. Zentralbl Bakteriol. 1960;179:289-297.
- Movila A, et al. Detection of tick-borne pathogens in ticks from migratory birds in the Baltic region of Russia. Med Vet Entomol. 2013;27(1):113-117.
- Obsomer V, et al. Spatial disaggregation of tick occurrence and ecology at a local scale as a preliminary step for spatial surveillance of tick-borne diseases: general framework and health implications in Belgium. Parasit Vectors. 2013;6:190.
- Labuda M, Nuttall PA. Tick-borne viruses. Parasitology. 2004;129 Suppl:S221-S245. doi:10.1017/s0031182004005220.
- Nowak-Chmura M, Siuda K. Ticks of Poland. Review of contemporary issues and latest research. Ann Parasitol. 2012;58(3):125-155.
- Moshkin MP, et al. Epidemiology of a tick-borne viral infection: theoretical insights and practical implications for public health. Bioessays. 2009;31(6):620-628.
- Hauser G, et al. Influence of climatic factors on Ixodes ricinus nymph abundance and phenology over a long-term monthly observation in Switzerland (2000-2014). Parasit Vectors. 2018;11(1):289.
- Jouda F, Perret JL, Gern L. Density of questing Ixodes ricinus nymphs and adults infected by Borrelia burgdorferi sensu lato in Switzerland: spatio-temporal pattern at a regional scale. Vector Borne Zoonotic Dis. 2004;4(1):23-32.
- Perret JL, et al. Darkness induces mobility, and saturation deficit limits questing duration, in the tick Ixodes ricinus. J Exp Biol. 2003;206(Pt 11):1809-1815.
- Perret JL, et al. Influence of saturation deficit and temperature on Ixodes ricinus tick questing activity in a Lyme borreliosis-endemic area (Switzerland). Parasitol Res. 2000;86(7):554-557.
- Obenchain FD, Galun R. Physiology of ticks. 1st ed. Current themes in tropical science. Oxford Oxfordshire; New York: Pergamon Press; 1982.
- Obenchain FD, Oliver JH Jr. Peripheral nervous system of the ticks, Amblyomma tuberculatum Marx and Argas radiatus Railliet (Acari: Ixodoidea). J Parasitol. 1976;62(5):811-817.
- Mao H, Kaufman WR. Profile of the ecdysteroid hormone and its receptor in the salivary gland of the adult female tick, Amblyomma hebraeum. Insect Biochem Mol Biol. 1999;29(1):33-42.
- Ribeiro JM. Role of saliva in blood-feeding by arthropods. Annu Rev Entomol. 1987;32:463-478.
- Ribeiro JM. Vector salivation and parasite transmission. Mem Inst Oswaldo Cruz. 1987;82 Suppl 3:1-3. doi:10.1590/ s0074-02761987000700003
- Ribeiro JM. Role of saliva in tick/host interactions. Exp Appl Acarol. 1989;7(1):15-20.
- Hoffmann A, et al. Isolation and characterization of a thrombin inhibitor from the tick Ixodes ricinus. Pharmazie. 1991;46(3):209-212.
- Balashov YS. Bloodsucking Ticks (Ixodoidea)—Vectors of Diseases of Man and Animals, in A Translation of ‘Bloodsucking Ticks (Ixodoidea)—Vectors of Diseases of Man and Animals’. 1972.
- Sonenshine DE. Biology of ticks. New York: Oxford University Press; 1991.
- Estrada-Peña A, Mihalca AD, Petney TN. Ticks of Europe and North Africa: A Guide to Species Identification. Cham: Springer International Publishing; 2017.
- Gage KL, et al. Climate and Vectorborne Diseases. Am J Prev Med. 2008;35(5):436-450.
- Gage KL, et al. Climate and Vectorborne Diseases. Am J Prev Med. 2008;35(5):436-450.
- Gaede K, et al. On the mechanism of water vapour sorption from unsaturated atmospheres by ticks. J Exp Biol. 1997;200 (Pt 10):1491-1498.
- Daniel M, et al. Predictive map of Ixodes ricinus highincidence habitats and a tick-borne encephalitis risk assessment using satellite data. Exp Appl Acarol. 1998;22 (7):417-433.
- Perez C, Rodhain F. [Biology of Ixodes ricinus L., 1758. I. Ecology; developmental cycle]. Bull Soc Pathol Exot Filiales. 1977;70(2):187-192.
- Perez C, Rodhain F. [Biology of Ixodes ricinus L., 1758. 2. Epidemiological consequences]. Bull Soc Pathol Exot Filiales. 1977;70(2):193-201.
- Talleklint L, Jaenson TG. Increasing geographical distribution and density of Ixodes ricinus (Acari: Ixodidae) in central and northern Sweden. J Med Entomol. 1998;35(4):521-526.
- Moran Cadenas F, et al. Phenology of Ixodes ricinus and infection with Borrelia burgdorferi sensu lato along a northand south-facing altitudinal gradient on Chaumont Mountain, Switzerland. J Med Entomol. 2007;44(4):683-693.
- Burri C, et al. Ixodes ricinus density and infection prevalence of Borrelia burgdorferi sensu lato along a North-facing altitudinal gradient in the Rhone Valley (Switzerland). Vector Borne Zoonotic Dis. 2007;7(1):50-58.
- Burda H, Šumbera R, Begall S. Microclimate in Burrows of Subterranean Rodents—Revisited. In: Subterranean Rodents. 2007:21-33.
- Gray JS, et al. Diapause in ticks of the medically important Ixodes ricinus species complex. Ticks Tick Borne Dis. 2016;7(5):992-1003.
- Korenberg EI. Seasonal population dynamics of ixodes ticks and tick-borne encephalitis virus. Exp Appl Acarol. 2000;24 (9):665-681.
- Naumov RL. [The longevity of the tick Ixodes ricinus (Acari: Ixodidae) in Central Russia]. Parazitologiia. 2006;40(4):384-395.
- Chitimia-Dobler L, et al. Repeated isolation of tick-borne encephalitis virus from adult Dermacentor reticulatus ticks in an endemic area in Germany. Parasit Vectors. 2019;12(1):90.
- Karbowiak G. The occurrence of the Dermacentor reticulatus tick—its expansion to new areas and possible causes. Ann Parasitol. 2014;60(1):37-47.
- Kiewra D, Czulowska A, Lonc E. Winter activity of Dermacentor reticulatus (Fabricius, 1794) in the newly emerging population of Lower Silesia, south-west Poland. Ticks Tick Borne Dis. 2016;7(6):1124-1127.
- Sands BO, Bryer KE, Wall R. Climate and the seasonal abundance of the tick Dermacentor reticulatus. Med Vet Entomol. 2021;35(3):434-441.
- Bartosik K, Wisniowski L, Buczek A. Abundance and seasonal activity of adult Dermacentor reticulatus (Acari: Amblyommidae) in eastern Poland in relation to meteorological conditions and the photoperiod. Ann Agric Environ Med. 2011;18(2):340-344.
- Schmuck HM, et al. Collection of immature Dermacentor reticulatus (Fabricius, 1794) ticks from vegetation and detection of Rickettsia raoultii in them. Ticks Tick Borne Dis. 2020;11(6):101543.
- Blaskovic D, Nosek J. The ecological approach to the study of tick-borne encephalitis. Prog Med Virol. 1972;14:275-320.
- Lichard M. [Remarks on the Occurrence and Ecology of the Tick Ixodes Trianguliceps Birula, 1895]. Biologia (Bratisl).1965;20:348-358.
- Alexandrov YV, Yagodinsky VN. The epidemiological significance of Ixodes persulcatus P. Sch. males in Russian spring-summer tick-borne encephalitis. J Hyg Epidemiol Microbiol Immunol. 1966;10(1):115-119.
- Bannova GG, et al. [Viral titers in Ixodes persulcatus ticks from various parts of a tick-borne encephalitis infected area]. Med Parazitol (Mosk). 1984;(1):34-36.
- Chumakov MP. Report on the isolation from Ixodes persulcatus ticks and from patients in western Siberia of a virus differing from the agent of tick-borne encephalitis. Acta Virol. 1963;7:82-83.
- Karbowiak G, et al. The transstadial persistence of tickborne encephalitis virus in Dermacentor reticulatus ticks in natural conditions. Acta Parasitol. 2016;61(1):201-203.
- Lickova M, et al. Dermacentor reticulatus is a vector of tickborne encephalitis virus. Ticks Tick Borne Dis. 2020;11 (4):101414.
- Riedl H, et al. [Isolation of the tick-borne encephalitis virus (TBE-virus) from the tick Haemaphysalis concinna Koch]. Arch Hyg Bakteriol. 1971;154(6):610-611.
- Kozuch O, Nosek J. Experimental transmission of tick-borne encephalitis (TBE) virus by Haemaphysalis concinna ticks. Acta Virol. 1980;24(5):377-379.
- Gresikova M, Noseck J. Isolation of tick-borne encephalitis virus from Haemaphysalis inermis ticks. Acta Virol. 1966;10(4):359-361.
- Kozuch O, Nosek J. The role of small rodents and hedgehogs in a natural focus of tick-borne encephalitis. Bull World Health Organ. 1967;36 Suppl(Suppl 1):
- Kozuch O, Nosek J. Alimentary infection of the hedgehog with tick-borne encephalitis (TBE) virus. Acta Virol.1964;8:284-285.
- Abdiyeva K, et al. Vectors, molecular epidemiology and phylogeny of TBEV in Kazakhstan and central Asia. Parasit Vectors. 2020;13(1):504.
- Karbowiak G, Kiewra D. New locations of Dermacentor reticulatus ticks in Western Poland: the first evidence of the merge in D. reticulatus occurrence areas? Wiad Parazytol.2010;56(4):333-336.
- Rubel F, et al. Geographical distribution of Dermacentor marginatus and Dermacentor reticulatus in Europe. Ticks Tick Borne Dis. 2016;7(1):224-233.
- Bakhvalova VN, et al. Tick-borne encephalitis virus diversity in ixodid ticks and small mammals in southwestern Siberia, Russia. Vector Borne Zoonotic Dis. 2016;16(8):541-549.
- Achazi K, et al. Rodents as sentinels for the prevalence of tick-borne encephalitis virus. Vector Borne Zoonotic Dis.2011;11(6):641-647.
- Achazi K, et al. Detection and differentiation of tick-borne encephalitis virus subtypes by a reverse transcription quantitative real-time PCR and pyrosequencing. J Virol Methods. 2011;171(1):34-39.
- Takeda T, et al. Isolation of tick-borne encephalitis virus from wild rodents and a seroepizootiologic survey in Hokkaido, Japan. Am J Trop Med Hyg. 1999;60(2):287-291.
- Ernek E, et al. Experimental infection of Clethrionomys glareolus and Apodemus flavicollis with tick-borne encephalitis virus. Acta Virol. 1963;7:434-436.
- Brandenburg PJ, et al. Seroprevalence of tick-borne encephalitis virus antibodies in wild rodents from two natural TBE foci in Bavaria, Germany. Pathogens. 2023;12(2).
- Hubalek Z, Rudolf I. Tick-borne viruses in Europe. Parasitol Res. 2012;111(1):9-36.
- Cisak E, et al. Prevalence of tick-borne encephalitis virus (TBEV) in samples of raw milk taken randomly from cows, goats, and sheep in eastern Poland. Ann Agric Environ Med.2010;17(2):283-286.
- Balogh Z, et al. Tick-borne encephalitis outbreak in Hungary due to consumption of raw goat milk. J Virol Methods. 2010;163(2):481-485
- Balogh Z, et al. Experimental infection of goats with tickborne encephalitis virus and the possibilities to prevent virus transmission by raw goat milk. Intervirology. 2012;55 (3):194-200.
- Cisak E, et al. Preliminary studies on the relationship between Ixodes ricinus activity and tick-borne infection among occupationally-exposed inhabitants of eastern Poland. Ann Agric Environ Med. 2001;8(2):293-295.
- Biernat B, Karbowiak G. Study on the occurrence of tickborne encephalitis virus RNA in European bison (Bison bonasus) eliminated at Bialowieza Primeval Forest (northeastern Poland) in 2005-2009. Ann Parasitol. 2014;60(2):99-102.
- Radda A, Kunz C, Hofmann H. [Demonstration of antibodies in sera of wild animals for the detection of foci of tick-borne encephalitis (TBE) virus in Lower Austria]. Zentralbl Bakteriol Orig. 1968;208(1):88-93.
- Wurm R, et al. Serological investigations of red foxes (Vulpes vulpes L.) for determination of the spread of tick-borne encephalitis in Northrhine-Westphalia. J Vet Med B Infect Dis Vet Public Health. 2000;47(7):503-509.
- Rieger MA, et al. Foxes as indicators for TBE endemicity—a comparative serological investigation. Zentralbl Bakteriol. 1999;289(5-7):610-618.
- Gresikova M, et al. Isolation of tick-borne encephalitis virus from the brain of a sick dog in Switzerland. Acta Virol.1972;16(1):88.
- Haemig PD, et al. Forecasting risk of tick-borne encephalitis (TBE): using data from wildlife and climate to predict next year’s number of human victims. Scand J Infect Dis. 2011;43 (5):366-372.
- Tonteri E, et al. Serological evidence of tick-borne encephalitis virus infection in moose and deer in Finland: sentinels for virus circulation. Parasit Vectors. 2016;9:54.
- Jemersic L, et al. Detection and genetic characterization of tickborne encephalitis virus derived from ticks removed from red foxes (Vulpes vulpes) and isolated from spleen samples of red deer (Cervus elaphus) in Croatia. Ticks Tick Borne Dis. 2014;5(1):7-13.
- Gerth HJ, et al. Roe deer as sentinels for endemicity of tickborne encephalitis virus. Epidemiol Infect. 1995;115(2):355-365.
- Kazarina A, et al. Detection of tick-borne encephalitis virus in I. ricinus ticks collected from autumn migratory birds in Latvia. Ticks Tick Borne Dis. 2015;6(2):178-180.
- Lommano E, et al. Tick-borne pathogens in ticks collected from breeding and migratory birds in Switzerland. Ticks Tick Borne Dis. 2014;5(6):871-882.
- Csank T, et al. Detection of West Nile virus and tick-borne encephalitis virus in birds in Slovakia, using a universal primer set. Arch Virol. 2016;161(6):1679-1683.
- Kupca AM, et al. Isolation and molecular characterization of a tick-borne encephalitis virus strain from a new tick-borne encephalitis focus with severe cases in Bavaria, Germany. Ticks Tick Borne Dis. 2010;1(1):44-51.
- Ponomareva EP, et al. Detection of Far-Eastern subtype of tickborne encephalitis viral RNA in ticks collected in the Republic of Moldova. J Vector Borne Dis. 2015;52(4):334-336.
- Ott D, et al. Tick-borne encephalitis virus (TBEV) prevalence in field-collected ticks (Ixodes ricinus) and phylogenetic, structural and virulence analysis in a TBE high-risk endemic area in southwestern Germany. Parasit Vectors. 2020;13(1):303.
- Paulsen KM, et al. Prevalence of tick-borne encephalitis virus in Ixodes ricinus ticks from three islands in north-western Norway. APMIS. 2015;123(9):759-764.
- Vikse R, et al. Geographical distribution and prevalence of tickborne encephalitis virus in questing Ixodes ricinus ticks and phylogeographic structure of the Ixodes ricinus vector in Norway. Zoonoses Public Health. 2020.
- Borde JP, et al. Tick-borne encephalitis virus infections in Germany. Seasonality and in-year patterns. A retrospective analysis from 2001-2018. PLoS One. 2019;14(10).
- Knap N, Avsic-Zupanc T. Factors affecting the ecology of tickborne encephalitis in Slovenia. Epidemiol Infect. 2015;143 (10):2059-2067.
- Süss J, et al. TBE incidence versus virus prevalence and increased prevalence of the TBE virus in Ixodes ricinus removed from humans. Int J Med Microbiol. 2006;296 Suppl 40:63-68.
- Waldenstrom J, et al. Migrating birds and tickborne encephalitis virus. Emerg Infect Dis. 2007;13(8):1215-1218.
- Korenberg EI, Kovalevskii YV. Main features of tick-borne encephalitis eco-epidemiology in Russia. Zentralbl Bakteriol. 1999;289(5-7):525-539.
- Korenberg EI, et al. Probability models of the rate of infection with tick-borne encephalitis virus in Ixodes persulcatus ticks. Folia Parasitol (Praha). 1992;39(1):85-92.
- Kim SY, et al. Molecular evidence for tick-borne encephalitis virus in ticks in South Korea. Med Vet Entomol. 2009;23(1):15-20.
- Daniel M, et al. Increased relative risk of tick-borne encephalitis in warmer weather. Front Cell Infect Microbiol. 2018;8:90.
- Topp AK, et al. Seroprevalence of tick-borne encephalitis virus in wild and domestic animals in northern Germany. Ticks Tick Borne Dis. 2023;14(6):102220.
- Mikryukova TP, et al. Surveillance of tick-borne encephalitis virus in wild birds and ticks in Tomsk city and its suburbs (Western Siberia). Ticks Tick Borne Dis. 2014;5(2):145-151.
- Gresikova M, et al. The role of birds in a natural focus of tickborne encephalitis. II. Experimental infection of great tits (Parus major L.) with tick-borne encephalitis virus. J Hyg Epidemiol Microbiol Immunol. 1962;6:339-342.
- Haut M, et al. The red fox (Vulpes vulpes) as sentinel for tickborne encephalitis virus in endemic and non-endemic areas. Microorganisms. 2020;8(11):1817. doi:10.3390/microorganisms8111817
- Tonteri E, et al. The three subtypes of tick-borne encephalitis virus induce encephalitis in a natural host, the bank vole (Myodes glareolus). PLoS One. 2013;8(12).
- Nuttall PA, et al. Adaptations of arboviruses to ticks. J Med Entomol. 1994;31(1):1-9.
- Chunikhin SP, et al. [Sexual transmission of the tick-borne encephalitis virus in ixodid ticks (Ixodidae)]. Parazitologiia. 1983;17(3):214-217.
- Fialova A, et al. Ixodes ricinus tick saliva modulates tick-borne encephalitis virus infection of dendritic cells. Microbes Infect. 2010;12(7):580-585.
- Danielova V, et al. Potential significance of transovarial transmission in the circulation of tick-borne encephalitis virus.Folia Parasitol (Praha). 2002;49(4):323-325.
- Swei A, et al. Patterns, drivers, and challenges of vector-borne disease emergence. Vector Borne Zoonotic Dis. 2020;20(3):159-170./li>
- Levin ML, et al. Minimal duration of tick attachment sufficient for transmission of infectious Rickettsia rickettsii (Rickettsiales: Rickettsiaceae) by its primary vector Dermacentor variabilis (Acari: Ixodidae): duration of rickettsial reactivation in the vector revisited. J Med Entomol. 2020;57(2):585-594.
- Nuttall PA. Displaced tick-parasite interactions at the host interface. Parasitology. 1998;116 Suppl:S65-S72. doi:10.1017/s003118200008495x.
- Ebel GD, Kramer LD. Short report: Duration of tick attachment required for transmission of Powassan virus by deer ticks. Am J Trop Med Hyg. 2004;71(3):268-271.
- Nosek J, et al. Localization of tick-borne encephalitis virus in alveolar cells of salivary glands of Dermacentor marginatus and Haemaphysalis inermis ticks. Acta Virol. 1972;16(6):493-497.
- Süss J. Epidemiology and ecology of TBE relevant to the production of effective vaccines. Vaccine. 2003;21 Suppl 1:S19-S35. doi:10.1016/s0264-410x(02)00812-5.
- Korenberg EI, et al. [Determination of the degree of infectiousness of adult ixodid ticks with the tick-borne encephalitis virus]. Vopr Virusol. 1986;31(3):319-321.
- Nosek J, et al. Peculiarities of tick-borne encephalitis virus reproduction in Haemaphysalis inermis ticks and their explants. Acta Virol. 1986;30(5):396-401.
- Nosek J, Grulich I. The relationship between the tick-borne encephalitis virus and the ticks and mammals of the Tribec mountain range. Bull World Health Organ. 1967;36 Suppl:31-47.
- Morozova OV, Panov VV, Bakhvalova VN. Innate and adaptive immunity in wild rodents spontaneously and experimentally infected with the tick-borne encephalitis virus. Infect Genet Evol. 2020;80:104187.
- Labuda M, et al. Amplification of tick-borne encephalitis virus infection during co-feeding of ticks. Med Vet Entomol. 1993;7(4):339-342.
- Labuda M, et al. Non-viraemic transmission of tick-borne encephalitis virus: a mechanism for arbovirus survival in nature. Experientia. 1993;49(9):802-805.
- Labuda M, et al. Efficient transmission of tick-borne encephalitis virus between cofeeding ticks. J Med Entomol. 1993;30(1):295-299.
- Labuda M, et al. Tick-borne encephalitis virus transmission between ticks cofeeding on specific immune natural rodent hosts. Virology. 1997;235(1):138-143.
- Il’enko VI, Gorozhankina TS, Smorodintsev AA. [Main regularities of transovarial transmission of tick-borne encephalitis virus by tick vectors]. Med Parazitol (Mosk). 1970;39(3):263-269.
- Slovak M, et al. Survival dynamics of tick-borne encephalitis virus in Ixodes ricinus ticks. Ticks Tick Borne Dis. 2014;5(6):962-969.
- Nuttall PA. Pathogen-tick-host interactions: Borrelia burgdorferi and TBE virus. Zentralbl Bakteriol. 1999;289(5-7):492-505.
- Danielova V, et al. Integration of a tick-borne encephalitis virus and Borrelia burgdorferi sensu lato into mountain ecosystems, following a shift in the altitudinal limit of distribution of their vector, Ixodes ricinus (Krkonose mountains, Czech Republic). Vector Borne Zoonotic Dis. 2010;10(3):223-230.
- Radda A, Hofmann H, Pretzmann G. Threshold of viraemia in Apodemus flavicollis for infection of Ixodes ricinus with tickborne encephalitis virus. Acta Virol. 1969;13(1):74-77.
- Rasnitsyn SP. [Evaluation of the importance of the transphasic and transovarian transmission for the preservation of a population of a causative agent]. Med Parazitol (Mosk). 1976;45(3):269-274.
- Chunikhin SP. [Experimental research on the ecology of the tick-borne encephalitis virus]. Vopr Virusol. 1990;35(3):183-188.
- Alekseev AN, Chunikhin SP. [The experimental transmission of the tick-borne encephalitis virus by ixodid ticks (the mechanisms, time periods, species and sex differences)]. Parazitologiia. 1990;24(3):177-185.
- Bakhvalova VN, et al. Vertical transmission of tick-borne encephalitis virus between generations of adapted reservoir small rodents. Virus Res. 2009;140(1-2):172-178.
- Dive I, et al. Tick-borne encephalitis virus (TBEV) infection in pregnancy: absence of virus transmission to the fetuses despite severe maternal disease—a case study. Ticks Tick Borne Dis. 2020;11(5):101491.
- Rosa R, Pugliese A. Effects of tick population dynamics and host densities on the persistence of tick-borne infections. Math Biosci. 2007;208(1):216-240.
- Rosa R, et al. Thresholds for disease persistence in models for tick-borne infections including non-viraemic transmission, extended feeding and tick aggregation. J Theor Biol. 2003;224(3):359-376.
- Martello E, et al. Systematic review on the non-vectorial transmission of Tick-borne encephalitis virus (TBEV). Ticks Tick Borne Dis. 2022;13(6):102028.
- Kerlik J, et al. Slovakia reports highest occurrence of alimentary tick-borne encephalitis in Europe: analysis of tick-borne encephalitis outbreaks in Slovakia during 2007-2016. Travel Med Infect Dis. 2018;26:37-42.
- Süss J. Tick-borne encephalitis in Europe and beyond—the epidemiological situation as of 2007. Euro Surveill. 2008;13(26).
- Jaenson TG, et al. Why is tick-borne encephalitis increasing? A review of the key factors causing the increasing incidence of human TBE in Sweden. Parasit Vectors. 2012;5:184.
- Kozuch O, Nosek J. Transmission of tick-borne encephalitis (TBE) virus by Dermacentor marginatus and D. reticulatus ticks. Acta Virol. 1971;15(4):334-336.
- Kriz B, Benes C, Daniel M. Alimentary transmission of tickborne encephalitis in the Czech Republic (1997-2008). Epidemiol Mikrobiol Imunol. 2009;58(2):98-103.
- Hudopisk N, et al. Tick-borne encephalitis associated with consumption of raw goat milk, Slovenia, 2012. Emerg Infect Dis. 2013;19(5):806-808.
- Brockmann SO, et al. A cluster of two human cases of tickborne encephalitis (TBE) transmitted by unpasteurised goat milk and cheese in Germany, May 2016. Euro Surveill. 2018;23(15).
- Lickova M, et al. Alimentary infections by tick-borne encephalitis virus. Viruses. 2021;14(1).
- Kerlik J, et al. Breast milk as a route of tick-borne encephalitis virus transmission from mother to infant. Emerg Infect Dis. 2022;28(5):1060-1061.
- Rizzoli A, et al. Forest structure and roe deer abundance predict tick-borne encephalitis risk in Italy. PLoS One. 2009;4(2).
- Pretzmann G, Radda A, Loew J. [Studies of a natural focus of the spring-summer meningoencephalitis (tick-borne encephalitis) in Lower Austria. 5. Additional investigations of the virus circulation in a natural focus]. Zentralbl Bakteriol Orig. 1964;194(4):431-439.
- Sandor AD, et al. Do the ticks of birds at an important migratory hotspot reflect the seasonal dynamics of Ixodes ricinus at the migration initiation site? A case study in the Danube Delta. PLoS One. 2014;9(2).
- Norman R, et al. Persistence of tick-borne virus in the presence of multiple host species: tick reservoirs and parasite mediated competition. J Theor Biol. 1999;200(1):111-118.
- Hartemink NA, et al. The basic reproduction number for complex disease systems: defining R0 for tick-borne infections. Am Nat. 2008;171(6):743-754.