Wilhelm Erber, Heinz-Josef Schmitt and Tamara Vuković Janković
Key points
- TBE is a flavivirus infection of the central nervous system (CNS), transmitted by ticks and in some instances by ingestion of unpasteurized milk.
- It is diagnosed in the forested belts of Northern Eurasia ranging from the UK, eastern France, The Netherlands and Norway down to Italy through central and Eastern Europe, Russia, Kazakhstan, and China to Japan.
- About 10,000 cases of TBE are reported annually, likely a significant underestimate as serological testing is more sporadic than complete and, in some countries, (like Japan) not even available.
- The European Centre for Disease Prevention and Control (ECDC) have put TBE on their list of notifiable diseases. Their case definition requires clinical symptoms of CNS infection plus virological or serological confirmation of the infection, usually by detection of specific immunoglobulins IgG and IgM.
- Vaccination against TBE is on the World Health Organization’s List of Essential Medicines. the safest and most effective medicines needed in a health system.
- Surveillance of TBE and the TBEV is incomplete. Reported incidences do not reflect actual risk since this fluctuates annually as a result of changes in exposure, vaccine uptake, intensity of case finding and reporting, climate factors, reservoir animals and ticks – just to mention the most relevant factors.
- For largely unknown reasons (including human behavior, improved diagnostics, or climate change) TBEV appears to be spreading north, east, west, even south and to higher altitudes to areas that were previously believed to be free of the virus.
Burden of disease and case definition
To date, tick-borne encephalitis virus (TBEV) foci have been identified in Europe, Siberia, far-eastern Russia, northern China, South Korea, and Japan. Up to 12,000 tick-borne encephalitis (TBE) cases are identified annually from countries where the disease is reportable. Mortality rates between 0.2% to 20% are reported, depending on region and perhaps on viral subtype.3 Severe long-term sequelae of TBE are well described both in children and in adults (see Chapters 5 and 6).
Because TBEV is present in reservoir animals in nature, eliminating or eradicating the disease is impossible. Thus, TBE is an important concern for the individual who becomes infected, but the disease is also of public health relevance, as acknowledged by the World Health Organization (WHO) in all position reports from 1983 to date (2011).3-5 Moreover, vaccination against TBE is on the World Health Organization’s List of Essential Medicines, the safest and most effective medicines needed in a healthcare system35. In addition, in 2012 the European Center for Disease Prevention and Control (ECDC) decided to add TBE to the list of mandatory notifiable diseases and provided for the first time ever a uniform disease case definition.2 (Table 1).
As the ECDC case definition and reporting have not been implemented around the globe and not even throughout Europe, data on the burden of disease from different countries are difficult to compare. Even if clear case definitions are provided and routinely implemented by local authorities, differences between countries exist regarding the classification of clinical diseases associated with TBEV infections. For example, Austria reports only “serologically proven hospitalized cases,” whereas the Czech Republic reports any case with “clinical and laboratory signs of aseptic meningitis/meningoencephalitis, not necessarily associated with hospitalization.”6
In addition to the use of different case definitions and case classifications, there is a lack of implementation of routine diagnostics in patients with encephalitis particularly with regard to detecting TBE. This is exemplified by the Polish experience: between 2004 and 2008, only 39% of the country’s hospitals had access to TBEV-serology. Therefore, a pilot project of enhanced surveillance for TBE was implemented in 2009.7 Testing for TBE in patients with signs of meningitis or encephalitis in the entire country doubled in 2009 compared with previous years, and 38 new endemic districts were identified. Seven of the new endemic districts were located far away from previously known endemic foci, most notably in the northwest of the country.
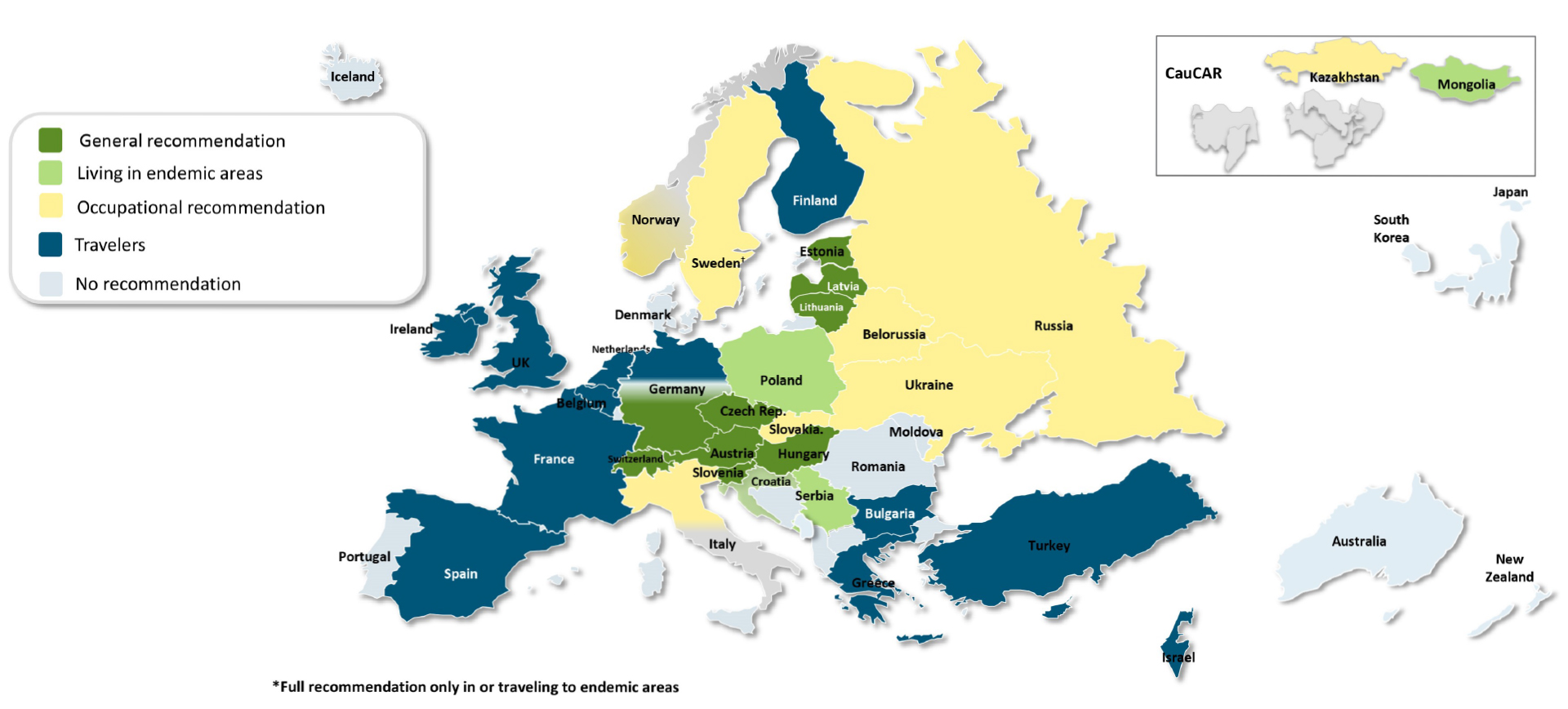
Finally, vaccine uptake may substantially modify the number of cases in a TBE risk area, as exemplified again by Austria, where in the last decade less than 100 cases are reported annually; this number was up to 700 cases annually before the introduction of a vaccination program. TBE vaccine uptake in Austria is around 84%. Neighboring countries with lower vaccine uptake continue to have increasing TBE case numbers.1 The following figures show countries with their respective current vaccination recommendations and vaccine reimbursement policies (Figure 1, Table 2).
Until 2018, only Austria has a national universal vaccination recommendation for the whole population, established a long time ago. Switzerland is the only other country that followed the same pathway, in February 2019 the entire country – except the cantons of Geneva and Ticino – is now defined as a TBE risk area by the Federal Office of Public Health and Vaccines Technical Committee). TBE vaccination is recommended for all persons in Switzerland (=6 years), who are tick-exposed and either live in a risk area or stay there temporarily; for children between 1 and 5 years, the situation is to be individually assessed. The entire Swiss population has a potential risk of exposure, depending on individual activity and mobility. In 2019, National Institute of Health Institute of Slovenia decided to partially fund the vaccination against TBE for children 3 years old and adults 49 year with three doses of the TBE vaccine (primary vaccination or booster). Previously unvaccinated adults 49 years old and children 3 years old, will be included in the vaccination program every year, thus gradually increasing the protection of the Slovenian population against TBE. (See chapter 12b Slovenia).
Recommendations in other countries, if they exist at all, are linked to certain conditions, e.g. predefined risk areas, age, or possible occupational exposure (Figure 1 and Table 2).
Overall, TBE surveillance in Europe is more sporadic than systematic, and TBE cases are likely underreported. For 2020, 24 EU/EEA countries reported 3,817 cases of tick-borne encephalitis, whereas the sum of all TBE cases outlined in the following country chapters results in 5,429 cases (Table 3).42 In the end, the real burden of disease from TBE remains unknown and the identification of TBEV endemic areas is far from being complete. With only inconsistent and incomplete scientific databases available, it is fair to conclude that the true TBEV disease burden is significantly underestimated.3,8
In this article we take the ECDC definition of TBE as a baseline (Table 1), requiring 1) clinically apparent disease of the central nervous system plus 2) at the same time a valid virological or serological documentation of current infection of the patient. However, in some countries “fever cases only”, if severe enough to be hospitalized and if laboratory confirmed, are also counted and reported as TBE cases. Reported numbers of TBE cases by country and by year are summarized in Table 3.
Table 1: TBE case definition by the ECDC4 ‘NA’= Not applicable
TICK-BORNE ENCEPHALITIS
- Clinical Criteria
Any person with symptoms of inflammation of the CNS (e.g. meningitis, meningoencephalitis, encephalomyelitis, encephaloradiculitis)
- Laboratory Criteria
Laboratory criteria for case confirmation:*
At least one of the following five:
– TBE specific IgM AND IgG antibodies in blood
– TBE specific IgM antibodies in CSF
– Sero-conversion or four-fold increase of TBE-specific antibodies in paired serum samples
– Detection of TBE viral nucleic acid in a clinical specimen,
– Isolation of TBE virus from clinical specimen
Laboratory criteria for a probable case:
– Detection of TBE-specific IgM-antibodies in a unique serum sample
- Epidemiological Criteria
Exposure to a common source (unpasteurized daily products)
Case Classification
- Possible case NA
- Probable case
Any person meeting the clinical criteria and the laboratory criteria for a probable case
OR Any person meeting the clinical criteria and with an epidemiological link
- Confirmed case
Any person meeting the clinical and laboratory criteria for case confirmation
*Serological results should be interpreted according to the vaccination status and previous exposure to other flaviviral infections. Confirmed cases in such situations should be validated by serum neutralization assay or other equivalent assays.
Figure 1: TBE vaccination recommendations
Click the image above to enlarge
Living in endemic areas – recommendation for residents in endemic areas
Occupational recommendation – limited recommendation for specific areas or outdoor activities, vaccination targets individuals in the most severely affected cohorts (e.g. forestry workers)
Travelers – recommendation only for travelers
Table 2: Country-specific recommendations and reimbursement for TBE vaccination (as of 2021)
| Country | Notifiable disease/ mandatory reporting system | Recommendation | Population | Reimbursed | Reimbursement type | Reimbursement details | ||
|---|---|---|---|---|---|---|---|---|
| Armenia | + | No recommendation | N | No reimbursement | No TBE vaccine registered | |||
| Australia | No recommendation | N | No reimbursement | No TBE vaccine registered | ||||
| Austria1,2,3,4 | All | Y | Partial reimbursement for all Austrians being vaccinated, and full reimbursement for special vacc. groups (eg, army, farmers) | Partial reimbursement for all Austrians being vaccinated, and full reimbursement for special vacc. groups (eg, army, farmers) | ||||
| Azerbaijan | – | No recommendation | N | No reimbursement | No TBE vaccine registered | |||
| Belarus3,5 | + | High Risk | TBE vaccine Moscow and Encepur available on the market | |||||
| Belgium3 | – | Travelers | Recommendation by the tropical institute for travelers to endemic regions and potential exposure (for travelers abroad to endemic areas) | N | No reimbursement | |||
| Bulgaria1,2,3,4 | – | Travelers | Recommendation for travelers to endemic | N | No reimbursement | No TBE vaccine registered | ||
| Croatia1,2,3,4 | + | At Risk + High Risk | Only recommended for residents in endemic areas and those visiting endemic areas (for recreation); forestry workers in the Koprivnica-Križevci region | N | Non-governmental reimbursement | Both registered, but only Austrian currently available | ||
| Czech Republic | + | All | Vaccination is recommended to all people permanently or temporarily residing in endemic areas with prevalence of TBE | Y | Non-governmental reimbursement | Contribution from preventive funds of health insurance companies only (approx. at the level of price of 1 dose of vaccine) | ||
| Denmark2,3 | – | |||||||
| Estonia1,2,3,4 | + | All | All individuals aged >1 y; Recommended for travelers visiting endemic areas | Y | Non- governmental reimbursement | reimbursement is available for the general population; Free for risk groups (foresters, irrigators, military personnel) vaccination is covered by the employer; both vaccines registered | ||
| Finland1,2,3,4 | + | Travelers | Travelers to endemic areas | |||||
| France1,2,3,4 | Travelers | Travelers to endemic areas | No reimbursement | |||||
| Germany1,2,3,4 | + | All | All individuals aged >1 y tick- exposed in RKI-defined (Robert Koch-Institut) TBE- risk areas, either they travel, live, and/or work there | Y | Reimb. for all those who live in, work in, or travel to RKI-defined ‘TBE-risk areas | Reimb. for all those who live in, work in, or travel to RKI- defined ‘TBE-risk areas (often even reimb. by insurances for travel to foreign endemic areas) | ||
| Georgia | + | No recommendation | N | No reimbursement | No TBE vaccine registered | |||
| Greece2,3 | + | Travelers | Travelers to endemic areas | |||||
| Hungary1,2,3,4 | + | All | Recommended for everyone | Y | Partial governmental reimbursement | Free for residents of highly endemic areas; Mandatory for people with extensive exposure to ticks in rural areas (eg, forestry workers and farmers since 1998, hikers and campers); German vaccine is reimbursed (25% of cost covered by National Health Insurance); both vaccines on the market | ||
| Iceland7 | – | No recommendation | ||||||
| Ireland8 | + | Travelers | A vaccination is the best way to prevent TBE for people living, working, or travelling in risk countries. You may consider having the TBE vaccination if: You are living in or planning to move to a risk country. Your work puts you at risk of TBE (for example, if you are a farmer, forestry worker or soldier). You are planning to travel to a risk area during late spring or summer and will be taking part in activities that put you at risk, such as camping, hiking, or bird watching. | N | No reimbursement | None | ||
| Israel9,10 | – | Travelers | Recommendation for travelers to the endemic area | Y | Partial governmental reimbursement | registered in Israel yet, and this is only OOP in traveler clinics and MOH clinics | ||
| Italy3,11 | – | High Risk | recommended to high- risk population groups (foresters, scouts, persons with hobbies or leisure activities potentially leading to tick exposure) in Veneto and in Trentino Alto Adige | |||||
| Kazakhstan3 | + | High Risk | Adults occupational (forest workers and soldiers) | Y | Partial governmental reimbursement | TBE vaccine Moscow is available on the market; FSME-immun is not registered | ||
| Kyrgyzstan | – | No recommendation | Y | No reimbursement | No TBE vaccine registered | |||
| Latvia1,2,5,6 | + | All | For children and adolescents living in endemic areas; strongly recommended for adults | Y | Partial governmental and private reimbursement | For children and adolescents living in endemic areas; for orphans/children without parental care – free of charge. 50% reimbursement for children (0-2y); 25% for pregnant women and women 42 days after delivery . Mandatory for high- risk groups and/or individuals expecting to have high occupational exposure (eg, forest workers and military personnel for whom vaccination is paid by employers); both vaccines registered. | ||
| Lithuania1,2,5,6 | + | All | Recommended for travelers and severely-affected cohorts | N | reimbursement | (e.g., forest workers); both vaccines registered | ||
| Luxembourg | ||||||||
| Malta5 | – | |||||||
| Mongolia20 | + | At Risk + High Risk | Adults occupational; residents and for tourists in provinces (Northern Mongolia- Selenge and Bulgan aimags) | Y | Partial governmental and private reimbursement | TBE vaccine Moscow and Chinese | ||
| Netherlands5,22 | – | Travelers | Persons with high-risk occupations in endemic areas; persons who go camping and hiking in nature reserves for longer than 2 days in the endemic regions of the Baltics, former Soviet Union, Kazakhstan, Mongolia, and Japan in the active tick season Persons who go camping and hiking in nature reserves for (cumulatively) longer than 4 weeks in the endemic areas of Central and Northern Europe in the active tick season | N | No reimbursement | |||
| Norway5 | + | |||||||
| Poland1,2,5,6 | + | At Risk + High Risk | Recommended for residents of endemic areas, particularly for military personnel, border guards, fire fighters, farmers, and tourists | Y | Non- governmental reimbursement | TBE vaccination is not universally reimbursed; Mandatory for forestry workers (since 1994) reimbursed by the employer; both vaccines registered | ||
| Portugal5 | – | |||||||
| Romania1,2,3,4 | + | No | No national TBE vaccination recommendations | N | No reimbursement | No national TBE vaccination policy; both vaccines registered | ||
| Russia3,5 | + | High Risk | Recommendations in the second part of NIP for endemic regions | Y | Partial governmental and private reimbursement | 3 locally produced Russian TBE vaccines, FSME-immun and Encepur are available on the market | ||
| Serbia14 | – | At Risk + High Risk | All aged >1 years of age residing or staying temporarily in endemic areas | N | No reimbursement | Vaccines are not registered | ||
| Slovakia1,2,3,4 | + | High Risk | Recommendation implemented only for high- risk occupational groups: forestry workers, farmers, surveyors, geologists, mountain hut and cableway staff, police officers, military personnel, and railway workers | Y | Non- governmental reimbursement | Implemented only for high- risk occupational groups; Mandatory for staff working in TBE testing laboratories; One private health insurance company (DOVERA) provide reimbursement of 3rd dose and second private health insurance company (UNION) provide reimbursement of 50% of each dose; both vaccines are available | ||
| Slovenia | + | All | Recommended for people living in or traveling to highly endemic areas, including children aged >1 y | Y | Partial governmental and private reimbursement | National TBE vaccination policy and recommendation implemented only for high- risk groups. Mandatory for high- risk workers; Mandatory for students at high risk, eg. forestry, wood processing (reimbursed within compulsory health insurance); both vaccines on the market Since March 2019- primary series are reimbursed for two cohorts children 3 years of age and adults 45-50 yrs of age | ||
| Spain2,3,16 | – | Travelers | Travelers to endemic areas | N | No reimbursement | Not applicable | ||
| Sweden | + | High Risk | The regional recommendation of Stockholm County is to vaccinate (3+1); no national TBE vaccination recommendations in different endemic settings or at different ages Travelers who intend to spend time in the outdoors, especially in the Stockholm archipelago, might want to get vaccinated | N | No reimbursement | No reimbursement is offered for TBE-vaccine | ||
| Switzerland4,19,20,21,22 | + | At Risk + High Risk | Adults and children >6 years of age residing or staying temporarily in endemic areas | Y | Full governmental reimbursement | Reimbursed for Swiss citizens (except region Genf and Tessin), for adults and children >6 years of age residing or staying frequently in endemic areas | ||
| Tajikistan | – | No recommendation | N | No reimbursement | No TBE vaccine registered | |||
| Turkey2,23 | – | Travelers | Individuals with high- risk activities (camping or working in farm and forest lands, adventurous journeys) and living in endemic countries | N | No reimbursement | |||
| UK24 | – | Travelers | Limited to travelers to high risk areas | N | No reimbursement | |||
| Ukraine3,5 | + | High Risk | N | No reimbursement | TBE vaccine Moscow and Encepur available on the market |
References (for Table 2)
- Zavadska D, Anca I, André F, et al. Hum Vaccin Immunother. 2013;9(2):362–374.
- ECDC Epidemiological situation of tick-borne encephalitis in the European Union and European Free Trade Association countries Available at: http://ecdc.europa.eu/en/publications/Publications/TBE-in-EU-EFTA.pdf#page=25 [Last accessed: September 2016]
- Kollaritsch H, et al. Vaccines and vaccination against tick-borne encephalitis. Exp Rev Vaccines. 2012;11(9):1103–1119.
- Mantke D, et al. A survey on cases of tick-borne encephalitis in European countries. Euro Surveill. 2008; 13(17).
- WHO International travel and health, Tick-borne Encephalitis. Available at: https://www.who.int/ith/2017-ith-chapter6_1-ru.pdf?ua=1 [Last accessed: February, 2018]
- Recommended procedure of the Czech Vaccinology Society for prevention and vaccination against tick-borne encephalitis (February 2016) Available online: http://www.vakcinace.eu/doporuceni-a-stanoviska [Last accessed: January 2018]
- Kanitz EE, et al. Variation in adult vaccination policies across Europe: An overview from VENICE network on vaccine recommendations, funding and coverage. Vaccine. 2012;30:5222– 5228.
- MoH Advisory committee report Ireland. Available at: https://www.hpsc.ie/a-z/vectorborne/publications/File,15592,en.pdf [Last accessed: April 2020]
- MoH Israel recommendations. Available at: http://www.health.gov.il/Subjects/vaccines/Vaccines_abroad/Documents/travel2013.pdf [Last accessed: April, 2020]
- MoH price list in the MOH clinics ILS 586. Available at: http://www.health.gov.il/PublicationsFiles/Price-vaccines-abroad.pdf [Last accessed: November, 2016]
- Rezza, et al. Surveillance and outbreak report. Italy Euro Surveill. 2015;20(40):pii=30034. DOI: http://dx.doi.org/10.2807/1560-7917.ES.2015.20.40.30034
- Khasnatinov M et al. (2010) Tick-borne encephalitis virus in Mongolia 14th International Congress on Infectious Diseases (ICID) Abstracts doi:10.1016/j.ijid.2010.02.449 LCR-guideline TBE, September 2015; access only granted to subscribers
- Recommendation of National immunization MoH Serbia. National Gazette of Republic of Serbia.2017; 82: 1-104. ISSN 0353-8389; CO-BISS.SR-ID 17264898
- Recommendations for vaccination against tick-borne meningoencephalitis, May 30th, 2019. Available at: https://www.nijz.si/sites/www.nijz.si/files/uploaded/kme-priporocila_za_cepljenje_proti_kme_2019_maj.pdf [Last accessed: March, 2020]
- MoH Spain Centro de Vacunación Internacional. Available at http://fundacionio.org/viajar/vacunas/vacuna%20encefalitis%20centroeuropea.html [Last accessed: April, 2020]
- Flower A. et al. Childhood encephalitis in Sweden: Etiology, clinical presentation and outcome. Eur j Paedriatr Neuro. 2008;12:484–490.
- Folkhälsomyndigheten, Public Health Authority Sweden Available at: https://www.folkhalsomyndigheten.se/the-public-health-agency-of-sweden/communicable-disease-control/vaccinations/vaccination-programmes/ [Last accessed: April, 2020]
- Schuler M. et al. Epidemiology of tick-borne encephalitis in Switzerland, 2005 to 2011. 2014;19(13):pii=20756. Available at: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=20756 [Last accessed: September, 2016]
- Federal Office of Public Health, Switzerland. Available at: https://www.bag.admin.ch/bag/de/home/zahlen-und-statistiken/zahlen-zu-infektionskrankheiten.exturl.html/aHR0cHM6Ly9tZWxkZXN5c3RlbWUuYmFnYXBwcy5jaC9pbmZyZX/BvcnRpbmcvZGF0ZW5kZXRhaWxzL2QvZnNtZS5odG1sP3dlYmdy/YWI9aWdub3Jl.html [Last accessed: March, 2020]
- Federal Vaccination program MoH Switzerland. Available at: https://www.bag.admin.ch/bag/de/home/krankheiten/krankheiten-im-ueberblick/fsme.html [Last accessed: April, 2020]
- MoH Switzerland. Available at: https://map.geo.admin.ch/mobile.html?layers=ch.bag.zecken-fsme-impfung&topic=ech&lang=de&bgLayer=ch.swisstopo.pixelkarte-farbe&layers_opacity=0.75 [Last accessed: April, 2020]
- Federal Office of Public Health Switzerland. Available at: https://lenews.ch/2019/01/04/vaccination-against-tick-borne-encephalitis-now-covered-by-swiss-health-insurance/ [Last accessed : February, 2018]
- Turkish Directorate General of Health for Border and Coastal Areas Available at: https://www.seyahatsagligi.gov.tr/Site/HastalikDetay/Kene-Kaynakli-Ensefalit [Last accessed: December 2017]
- UK Green book chapter 31 Available at: https://www.gov.uk/government/publications/tick-borne-encephalitis-the-green-book-chapter-31 [Last accessed: April, 2020].
Table 3: TBE cases by year and country
| Country | 1991 | 1992 | 1993 | 1994 | 1995 | 1996 | 1997 | 1998 | 1999 | 2000 | 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Albania | ||||||||||||||||||||||||||||||||
| Austria1 | 128 | 84 | 102 | 178 | 109 | 128 | 99 | 62 | 41 | 60 | 54 | 60 | 82 | 54 | 100 | 84 | 45 | 87 | 79 | 62 | 113 | 52 | 98 | 80 | 64 | 89 | 116 | 154 | 108 | 216 | 128 | 179 |
| Belarus2 | 23 | 61 | 18 | 53 | 44 | 46 | 108 | 82 | 66 | 88 | 91 | 108 | 122 | 109 | 119 | 77 | 141 | 142 | 135 | 171 | 108 | 108 | No data | |||||||||
| Belgium3-6 | 2 | 3 | 1 | 1 | 3 | 2 | 0 | 3 | 2 | 2 | ||||||||||||||||||||||
| Bosnia and Hercegovina7 | 1 | 2 | 5 | 0 | N/A | N/A | ||||||||||||||||||||||||||
| Bulgaria2 | 2 | 0 | 0 | 1 | 0 | 0 | 2 | 0 | 1 | 0 | 1 | 2 | 1 | 0 | ||||||||||||||||||
| China | ||||||||||||||||||||||||||||||||
| Croatia7-9 | 60 | 27 | 76 | 87 | 59 | 57 | 25 | 24 | 26 | 18 | 27 | 30 | 36 | 38 | 28 | 20 | 12 | 20 | 44 | 36 | 26 | 45 | 44 | 42 | 25 | 6 | 10 | 24 | 14 | 15 | 4 | 23 |
| Czech Republic10 | 356 | 337 | 618 | 619 | 727 | 571 | 412 | 422 | 490 | 709 | 633 | 647 | 606 | 507 | 642 | 1028 | 546 | 633 | 816 | 589 | 861 | 573 | 625 | 410 | 355 | 565 | 687 | 715 | 774 | 855 | 594 | 710 |
| Denmark2 | 2 | 2 | 3 | 4 | 3 | 3 | 1 | 4 | 8 | 2 | 2 | 1 | 2 | 2 | 4 | 1 | 1 | 3 | 1 | 1 | 1 | 0 | 4 | 5 | 5 | 7 | 5 | |||||
| Estonia11 | 68 | 163 | 166 | 177 | 175 | 177 | 404 | 387 | 185 | 272 | 215 | 90 | 237 | 182 | 164 | 171 | 140 | 90 | 179 | 201 | 250 | 178 | 113 | 84 | 116 | 81 | 87 | 85 | 83 | 70 | 80 | 138 |
| Finland12 | 14 | 25 | 16 | 23 | 8 | 19 | 16 | 12 | 42 | 33 | 38 | 16 | 29 | 16 | 18 | 20 | 23 | 25 | 38 | 43 | 39 | 38 | 47 | 68 | 61 | 82 | 79 | 69 | 91 | 148 | 124 | |
| France2 | 1 | 1 | 4 | 3 | 4 | 1 | 2 | 2 | 5 | 5 | 8 | 4 | 3 | 8 | 4 | 10 | 6 | 6 | 2 | 3 | 8 | 4 | 4 | 10 | 11 | 29 | 18 | 24 | 24 | 68 | 31 | 31 |
| Germany13-15 | 44 | 142 | 118 | 306 | 226 | 114 | 211 | 148 | 115 | 133 | 255 | 239 | 277 | 274 | 432 | 544 | 239 | 289 | 313 | 260 | 424 | 195 | 420 | 264 | 221 | 353 | 485 | 582 | 443 | 717 | 421 | 555 |
| Greece16 | 1 | 1 | ||||||||||||||||||||||||||||||
| Hungary2 | 299 | 190 | 339 | 264 | 234 | 246 | 102 | 74 | 69 | 54 | 55 | 80 | 114 | 89 | 54 | 57 | 63 | 55 | 70 | 50 | 43 | 44 | 53 | 31 | 24 | 19 | 16 | 32 | 18 | 18 | 6 | 29 |
| Italy2 | 0 | 2 | 2 | 8 | 6 | 8 | 8 | 11 | 5 | 12 | 24 | 9 | 17 | 32 | 25 | 44 | 21 | 26 | 34 | 21 | 26 | 34 | 42 | 22 | 14 | 53 | 24 | 40 | 37 | 55 | 14 | 40 |
| Japan18 | 1 | 1 | 2 | 1 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||
| Kazakhstan2 | 20 | 19 | 12 | 17 | 22 | 30 | 43 | 38 | 60 | 44 | 35 | 55 | 30 | 50 | 49 | 33 | 32 | 34 | 49 | 30 | 40 | 33 | 27 | 28 | 49 | 48 | 34 | 46 | 35 | 31 | 24 | 32 |
| Kyrgyzstan | N/A | N/A | ||||||||||||||||||||||||||||||
| Latvia19 | 227 | 287 | 791 | 1366 | 1341 | 736 | 874 | 1029 | 350 | 544 | 303 | 153 | 365 | 251 | 142 | 170 | 129 | 125 | 210 | 306 | 280 | 232 | 207 | 139 | 132 | 213 | 176 | 152 | 211 | 210 | 249 | 240 |
| Lithuania20 | 14 | 17 | 198 | 284 | 427 | 310 | 645 | 548 | 171 | 419 | 298 | 168 | 763 | 425 | 243 | 462 | 234 | 220 | 605 | 612 | 365 | 495 | 501 | 353 | 336 | 633 | 474 | 384 | 711 | 679 | 365 | 377 |
| Moldova | 0 | No data | No data | |||||||||||||||||||||||||||||
| Mongolia2 | 5 | 6 | 52 | 12 | 8 | 9 | 13 | 6 | 15 | 7 | 40 | 52 | 62 | 32 | 19 | 20 | 5 | 8 | ||||||||||||||
| Netherlands21-23 | 0 | 0 | 0 | 2 | 1 | 2 | 2 | 5 | 2 | 2 | ||||||||||||||||||||||
| Norway24 | 1 | 1 | 1 | 0 | 2 | 1 | 4 | 4 | 5 | 13 | 11 | 10 | 11 | 14 | 7 | 6 | 13 | 9 | 12 | 16 | 26 | 35 | 41 | 71 | 90 | |||||||
| Poland25 | 4 | 8 | 241 | 181 | 267 | 259 | 201 | 208 | 208 | 170 | 210 | 126 | 339 | 262 | 177 | 317 | 233 | 202 | 351 | 294 | 221 | 190 | 227 | 195 | 149 | 284 | 283 | 197 | 265 | 158 | 210 | 445 |
| Romania2,26 | 8 | 4 | 3 | 3 | 3 | 1 | – | 0 | No data | No data | ||||||||||||||||||||||
| Russia27-29 | 5194 | 6239 | 7571 | 5640 | 5935 | 10371 | 6804 | 7531 | 10011 | 6010 | 6569 | 5231 | 4773 | 4178 | 4593 | 3433 | 3142 | 3140 | 3141 | 3094 | 3533 | 2716 | 2236 | 1978 | 2304 | 2035 | 1934 | 1727 | 1781 | 989 | 1015 | 1969 |
| Serbia2,30 | 1 | 6 | 1 | 4 | 4 | 1 | 5 | 13 | 0 | 0 | No data | |||||||||||||||||||||
| Slovakia31 | 24 | 16 | 51 | 60 | 89 | 82 | 76 | 54 | 63 | 92 | 75 | 62 | 74 | 70 | 50 | 91 | 57 | 79 | 76 | 90 | 108 | 107 | 162 | 117 | 88 | 174 | 75 | 156 | 161 | 185 | 96 | 203 |
| Slovenia32,33 | 118 | 80 | 197 | 531 | 157 | 406 | 274 | 137 | 150 | 196 | 260 | 262 | 282 | 199 | 297 | 372 | 199 | 251 | 304 | 166 | 247 | 164 | 309 | 100 | 62 | 83 | 102 | 153 | 111 | 187 | 62 | 126 |
| South Korea | 0 | N/A | No data | |||||||||||||||||||||||||||||
| Sweden34 | 68 | 84 | 48 | 116 | 67 | 45 | 74 | 65 | 53 | 133 | 128 | 104 | 101 | 174 | 126 | 161 | 181 | 224 | 210 | 174 | 284 | 287 | 209 | 178 | 268 | 238 | 391 | 385 | 358 | 274 | 534 | 465 |
| Switzerland35 | 37 | 66 | 44 | 97 | 60 | 62 | 123 | 68 | 112 | 89 | 96 | 52 | 114 | 131 | 204 | 238 | 105 | 119 | 112 | 96 | 170 | 96 | 202 | 108 | 122 | 200 | 273 | 369 | 259 | 448 | 285 | 391 |
| Tunisia | 0 | No data | ||||||||||||||||||||||||||||||
| Ukraine36,37 | 12 | 28 | 4 | 8 | 7 | 4 | 7 | 8 | 3 | 10 | 3 | 3 | 6 | 3 | 6 | 4 | 5 | 2 | 2 | No data | No data | |||||||||||
| United Kingdom38,39 | 2 | 0 | 2 |
References (for Table 3)
- VIRUSEPIDEMIOLOGISCHE INFORMATION Austria 2013-2022 Available at: https://www.virologie.meduniwien.ac.at/wissenschaft-forschung/virus-epidemiologie/virusepidemiologische-information/2023/ [Last accessed: March, 2023]
- Dobler, G.; Erber, W.; Schmitt H.J.: TBE-The Book, Global Health Press, Singapore 2017 (ISBN: 978-981-1903-3).URL: https://tbenews.com/tbe [Last accessed: March 2020]
- Scientific Institute of Public Health of Belgium Service Épidémiologie des maladies infectieuses 2013-2014 Available at: https:// epidemio.wiv-isp.be/ID/reports/Zoonoses%20et%20maladies%20%c3%a0%20transmission%20vectorielle.%20Rapport%20annuel% 202013%20et%202014.pdf [Last accessed: March, 2020]
- Lernout, M. Van Esbroeck Surveillance épidémiologique de l’encéphalite à tiques TBEV – 2017
- Carole Schirvel (surveillance.sante@aviq.be) et Javiera Rebolledo (javiera.rebolledo@sciensano.be);FLASH Maladies infectieuses – Nov 2018 – N°11
- Siensano Epidemiology of infectious diseases. Available at: https://epidemio.wiv-isp.be/ID/reports/Zoonoses%20et%20maladies%20vectorielles%202018.pdf [Last accessed: March, 2020]
- Institute of Public Health FBIH http://www.zzjzfbih.ba/izdavastvo/publikacije/ [Last accessed: October, 2016]
- Markovinovic L, et al. An outbreak of tick-borne encephalitis associated with raw goat milk and cheese consumption, Croatia, 2015. Infection. 2016; 44: 661–665.
- Personal communication – Rok Čivljak, University of Zagreb School of Medicine, Zagreb, Croatia
- National Institute of Publi Health, Czech Republic. Available at: http://www.szu.cz/publikace/data/2019/vyskyt-vybranych-hlasenych-infekci-v-ceske-republice?lang=1#article [Last accessed: March, 2023]
- Estonia Available at: https://www.terviseamet.ee/et/nakkushaigused-menuu/tervishoiutootajale/nakkushaigustesse-haigestumine [Last accessed: March, 2023]
- National Registry of Infectious diseases of Finland. Available at: https://sampo.thl.fi/pivot/prod/fi/ttr/shp/fact_shp?row=area-12260&column=time-12059&filter=reportgroup-12194 [Last accessed: May, 2023]
- Robert Koch Institute Available at: https://www.rki.de/DE/Content/Infekt/Jahrbuch/jahrbuch_node.html [Last accessed: May, 2023]
- Robert Koch-Institut, Aktuelle Statistik meldepflichtiger Infektionskrankheiten, Deutschland, 44. Woche 2018 (Datenstand: 21. November 2018). Epid Bull. 2018;47:510.
- Survstat@RKI. Available at https://survstat.rki.de/Content/Query/Create.aspx [Last accessed May, 2023]
- Hellenic Center for Disease Control & Prevention. Available at: http://www.keelpno.gr/en-us/home.aspx [Accessed: June 2018]
- National Public Health and Medical Officer Service, Hungary Available at: https://www.antsz.hu/felso_menu/temaink/jarvany/Fertozo_betegsegek/Fertozo_eves_jelentesk [Last accessed: March, 2022]
- Yoshii K. et al. Tick-borne encephalitis in Japan, Republic of Korea and China. Emerg Microbes Infect. 2017 Sep 20;6(9):e82.
- Ministry of Health of the Republic of Latvia Available at: https://www.spkc.gov.lv/lv/epidemiologijas-bileteni [Last accessed: March, 2023]
- Ministry of Health of the Republic of Lithuania Available at: https://nvsc.lrv.lt/lt/uzkreciamuju-ligu-valdymas/statistika-apie-uzkreciamasias-ligas [Last accessed: March, 2023]
- Dutch National Institute for Public Health (RIVM) July 2016. Available at: https://www.rivm.nl/en/news/first-patient-infected-by-tick-borne-encephalitis-virus [Last accessed: March, 2020]
- De Graaf JA, Reimerink JHJ, Voorn GP, bij de Vaate EA, de Vries A, Rockx B, Schuitemaker A, Hira V. First human case of tick-borne encephalitis virus infection acquired in the Netherlands, July 2016. Euro Surveill. 2016;21(33):pii=30318. doi: 10.2807/1560-7917.ES.2016.21.33.30318
- Fanoy, E. TBE acquired in The Netherlands. Infectious diseases. 2017. 28(7): p: 205.
- Norwegian Surveillance System for Communicable Disease (MSIS)Available at: Reference: https://www.fhi.no/nettpub/smittevernveilederen/sykdommer-a-a/skogflattencefalitt-tbe-virusinfeks/ [Last accessed: March, 2023]
- National Institute of Public Helath, Poland (www.pzh.gov.pl) Available at: http://wwwold.pzh.gov.pl/oldpage/epimeld/index_a.html#01 [Last accessed: March, 2020]
- National Institute of Public Health, Romania. Available at: http://www.insp.gov.ro/ [Last accessed: August 2017]
- Noskov AK, et al. Tick-Borne Virus Encephalitis in the Russian Federation: Features of Epidemic Process in Steady Morbidity Decrease Period. Epidemiological Condition in 2016 and the Forecast for 2017. Problems of Particularly Dangerous Infections. 2017;1:37–43. (In Russ.). doi: 10.21055/0370-1069-2017-1-37-43 http://journal.microbe.ru/jour/article/view/367
- National surveillance on Infectious disease, Russia. Available at: http://rospotrebnadzor.ru [Last accessed: March, 2023]
- iMonitoring. Available at: https://www.iminfin.ru/areas-of-analysis/health/perechen-zabolevanij [Last accessed: May, 2023]
- Poluga J. et al. Tick-borne encephalitis in Serbia: A case series. J Infect Dev Ctries. 2019; 13(6):510-515.
- Epidemiological information system, Slovakia. Available at: http://www.epis.sk/AktualnyVyskyt/PrenosneOchorenia/Grafy/Tyzden52.aspx [Last accessed: December, 2018]
- NIPH, Available at: http://www.nijz.si/sl/tedensko-spremljanje-lymske-borelioze-in-klopnega-meningoencefalitisa [Last accessed: March, 2020]
- Slovenian Environment Agency. Available at: https://kazalci.arso.gov.si/sl/content/prijavljeni-primeri-lymske-borelioze-klopnega-meningoencefalitisa-v-sloveniji-2 [Last accessed: May, 2023]
- Statistical database, Sweden. Available at: https://www.folkhalsomyndigheten.se/sok/?q=f%C3%A4stingburen+encefalit [Last accessed March, 2023]
- Federal Office of Public Health, Switzerland. Available at: https://www.bag.admin.ch/bag/de/home/zahlen-und-statistiken/zahlen-zu-infektionskrankheiten.exturl.html/aHR0cHM6Ly9tZWxkZXN5c3RlbWUuYmFnYXBwcy5jaC9pbmZyZX/BvcnRpbmcvZGF0ZW5kZXRhaWxzL2QvZnNtZS5odG1sP3dlYmdy/YWI9aWdub3Jl.html [Last accessed: March, 2023]
- Amicizia D, et al. Epidemiology of tick-borne encephalitis (TBE) in Europe and its prevention by available vaccines. Hum Vaccin Immunother. 2013; 9(5): 1163–1171.
- Ben I. and Lozynskyi I. Prevalence of Anaplasma phagocytophilum in Ixodes ricinus and Dermacentor reticulatus and Coinfection with Borrelia burgdorferi and Tick-Borne Encephalitis Virus in Western Ukraine. Vector Borne and Zoonotic Diseases (Larchmont, N.Y.), 18 Jun 2019, 19(11):793-80.
- Mansbridge CT, Osborne J, Holding M, Dryden M, Aram M, Brown K, Sutton J. Autochthonous tick-borne encephalitis in the United Kingdom: A second probable human case and local eco-epidemiological findings. Ticks Tick Borne Dis. 2022 Jan;13(1):101853. doi: 10.1016/j.ttbdis.2021.101853. Epub 2021 Oct 10. PMID: 34670189.
- UK Health Security Agency. Available at: https://www.gov.uk/guidance/tick-borne-encephalitis-epidemiology-diagnosis-and-prevention [Last accessed: May, 2023]
TBE risk areas
While the challenges of detecting TBE cases in Europe have been described above, here we look at the collection of data to identify TBEV-endemic areas. Again, several methods are employed in different countries for epidemiological mapping:9
- testing of ticks and animal reservoirs for the presence of TBEV (especially by molecular diagnostic techniques);
- seroprevalence studies of human as well as animal populations exposed to ticks; and
- description of clinical cases with verifiable tracking of the place where the infection was acquired.
Each of these methods gives only a part of the complete picture. Some countries report the geographic prevalence of TBE based on the incidence of human cases only. However, this type of information does not give a clear picture on TBE endemic areas because often the exact place of TBE infection cannot be determined with certainty. Thus some TBE cases are ‘lost’ for surveillance and reporting for the location where the infection was acquired. Overall, data on TBEV distribution are incomplete, heterogeneous between the different countries, and sometimes even inconsistent for the same country.
In the end, estimating the risk of infection by the TBEV in a specific (endemic or non-endemic) area is impossible for several reasons.
- The epidemiology of TBE is the result of a complex interaction between reservoir animals, birds, ticks, plants, climate, weather, and human behavior (including vaccine uptake; see Chapters 3 and 13 for details). These variables change annually and unpredictably resulting in great annual differences in case numbers, as is well demonstrated in Chapter 12b (country-specific summaries) as well as in Table 2 above.
- A high local vaccine uptake may result in a low disease incidence, whereas the incidence in the unvaccinated (e.g., a traveler) may be much higher than the reported risk in the local population indicates.
- TBEV exists in microfoci, i.e. the virus often is detectable in small areas only, whereas the surrounding areas are TBEV-free.
Considering these points, the prevalence of TBEV in ticks obviously can vary considerably, even within 1 country or 1 area within an endemic region. For instance, in some highly endemic areas, TBEV prevalence in ticks reaches 20– 40%, but in other areas it can be as low as 0.1–0.5%3 (see Chapter 11). Or: a highly TBE endemic area may have a very low population size, so no TBE cases or only low incidence numbers of TBE cases are identified. Thus, the area may appear to be TBE-free whereas hikers in the area may have a high risk.
Finland for example is the eighth-largest country in Europe and the most sparsely populated country in the European Union (Population density is 18 inhabitants per square kilometer. This is the third-lowest population density of any European country). The majority of the population lives in the central and southern parts of the country. However, according to monitoring data for 2015–2019, the calculated incidence of tick-borne encephalitis in 2019 is as high as 53 per 100.000 inhabitants in the municipality of Pargas, 42 in Simo, 20 in Kustavi, and 30 on the island of Åland. Recommendations per municipality are based on human incidence numbers exclusively and do not consider those many municipalities where there are only few people living 40.
With this in mind, the TBE-incidence of a country alone is not an adequate measure for the individual risk to acquire TBE. Moreover, to date there is no commonly accepted definition to characterize “TBE risk areas”. To address this problem in a transparent and scientific way the country surveys listed in Chapter 12b of THE TBE BOOK are based on a proposal by ECDC10for assessing the risk for arbovirus infections in general.
- The key point from this10 is that “… any area where the chances of transmission of an arthropod-borne disease to humans are higher than nil is a risk area.” This definition is compelling as it refrains from requiring any specific level of risk (which can be small or large), like incidence data, which vary from year to year even for the same region.
- A predisposed area is a risk area where existing conditions might facilitate the transmission of an to humans, but the respective pathogen has not been detected.
- An imperiled area is a risk area where the pathogen has been detected in vectors, or transmission of the pathogen to animals or humans has been detected indirectly (by serology).
- An affected area is a risk area, where human TBE disease cases have occurred either sporadically or in a timewise restricted matter.
- An endemic area is a risk area where recurrent transmission of TBE to humans is taking place over several seasonal cycles.
In order to assign an arbovirus-risk based on the ECDC definition10 an area must be accurately determined geographically and by biological and epidemiological findings (surveillance of human and animal cases, field investigation etc.) in order to avoid misunderstandings and imprecision. This however is NOT the case with TBE, as the quality of surveillance and reporting is significantly different among countries and data cannot be simply compared. Therefore, the ECDC classification is by no means a risk assessment, but rather a way to grade available evidence.
In South Korea, TBEV has been detected in ticks, but no single human case has been identified to date. In Japan, only 1 case had been confirmed by 1993, and 4 other human TBE cases were identified between 2016 and 2018. In The Netherlands 3 autochthonous TBE cases were identified in 2017 and a total of three TBE foci have been identified so far.11,12 In Belgium, circulation of the virus in wild life has been documented, and the possibly first two autochthonous human TBE disease cases were reported.36,37 Recently, circulation of the TBEV in wild life as well as one possible indigenous human TBE case has been reported from the UK38,39 With this in mind, it remains unknown if travelers to South Korea have any risk for TBE if exposed to ticks in this country, whereas clearly, at least some parts of The Netherlands, Belgium, Japan and the UK are now TBEV-affected areas at least, and they may become endemic in case universal testing is applied.
Physicians, travelers, or the public in general may refer to the respective country in Chapter 12b of TBE TBE BOOK to see the number of reported cases by year, the local vaccine uptake (as available), and other key information to judge on possible risks – bearing in mind the limitations, and the accuracy of surveillance in the given country, and reporting mentioned herein.
It is the task of local authorities to define “TBE-areas” and decide on recommendations for vaccination either for all persons living in an area or for special risk groups. As the epidemiology of TBE changes annually it has to continuously reevaluated.
See Chapter 12b for evidence of TBEV/TBE in individual countries.
See Chapter 12c: Risk map for TBE virus.
Areas without confirmed TBE risk
Lack of circulation of the TBEV in Eurasia has been confirmed for Spain, Portugal, The Republic of Ireland, Luxembourg, Georgia, Albania, Kosovo, Macedonia, Montenegro, Greece, and Turkey as well as for some other parts of the continent.
First reports of TBEV seropositivity for TBEV in Spain were published in animals in Extremadura in 2003 and in Andalusia in 2014, and neutralizing antibodies against TBEV were recently detected in a horse on the island of Mallorca (off the eastern coast of Spain).13,14 Nonetheless these data are difficult to interpret as they could be due to cross-reactivity with IgG directed against closely related viruses of the same serogroup. Indeed, the louping ill virus, a member of the TBEV serocomplex, has already been detected in ticks and livestock in Spain.
An investigation in Turkish blood donors in Zonguldak in the Black Sea region has shown one TBEV positive sample by NT (PRNT).15 However, cross-reactions in commercial serological tests as well as by PRNT cannot be entirely ruled out. Recently IgM but no IgG antibodies to the TBEV were detected by ELISA in five children in Turkey.16 This constellation is highly implausible and indicates possibly nonspecific cross reactions as well. More data on TBE are required in order to confirm the existence of the TBEV in Turkey.
Eltari reported a total of 82 TBE cases from 1983 to 1990 in Albania. No further data are available after 1990.43,44 There is only one report from Bosnia mentioning TBE cases.45
In a serological study in Northern Greece serum samples from 921 apparently healthy individuals were investigated for the presence of TBEV antibodies. According to the authors two percent of the general population was found to be TBE-seropositive in their test system.17 However as TBEV is not endemic in Greece these findings may well result from cross-reactivity to Greek Goat Encephalitis Virus. Nonetheless two imported ΤΒΕ cases confirmed by neutralization test were reported to TESSy-ECDC, one in 2014 and a second in 2016.18
Recent data definitely confirmed the presence of TBEV in Northern Italy (see country chapter). Older data show up 14 human TBE cases and two virus isolations between 1975 and 2004 in the Toscana region however, no additional TBE case has been reported ever since. Still, a seroprevalence study in hunters and wild boar breeders in Turin Province and in particular in the Susa valley showed an about 5% seroprevalence by ELISA and amongst low risk individuals seroprevalence was below 2%.19
In Afghanistan, a study showed 23.4% seroprevalence and 20 human cases of IgM positivity; however, cross-reaction with Royal Farm virus cannot be excluded.46
In Georgia, 7% of acute febrile patients showed TBEV seropositivity.47
Most recently a study investigated the possible circulation of TBEV in Northern Iran, where climatic conditions, presence of Ixodes ticks, and variability of mammalian hosts might contribute to TBEV establishment. Anti-TBEV IgG antibody positive ELISA results were reported41; however, no confirmatory test was done, and cross-reactivity among flaviviruses is highly plausible.
Within the Central Asian countries there are reports of TBE in Kazakhstan and Kyrgyzstan (see country chapters), the only other single report without any further details is Turkmenistan.48
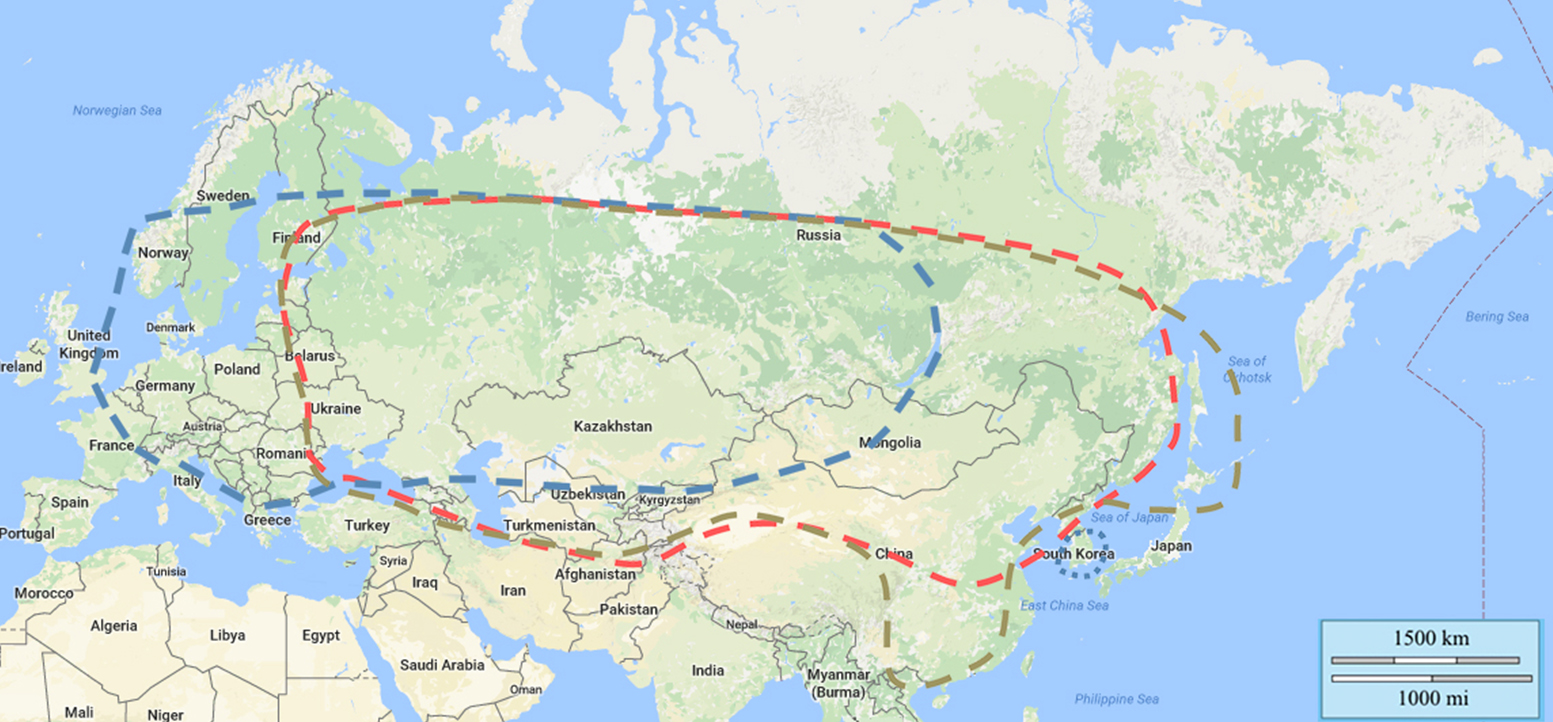
Figure 3: Distribution of TBEV subtypes by country
Click the image above to enlarge
Distribution of TBEV subtypes:
| TBEV-Eu | dotted blue line: prevails Europe, virus isolates in Siberia also, most eastern virus isolation Lake Baikal |
| TBEV-Sib | dotted red line: prevails Siberia and Ural region, most western virus isolation Baltics and Moldavia, most eastern virus isolation far eastern region of Russia |
| TBEV-Fe | dotted brown line: prevails far eastern region of Russia, most western virus isolation Baltics and Moldavia, most eastern virus isolation Hokkaido, Japan |
| Islands of unusual TBEV subtype distribution are reported in South Korea (TBEV-EU) | |
TBEV subtype and vector distribution
Three main TBEV subtypes have been described based on their main distribution pattern and sequence similarity: the European virus (previously CEE virus, Central European encephalitis virus; TBEV-EU), the Far Eastern virus (previously RSSE virus; TBEV-FE), and the Siberian virus (previously west Siberian virus; TBEV-Sib). In addition to the 3 primary TBEV subtypes, there is a fourth accepted subtype, designated as ( Baikalian subtype (TBEV-BKL) with the prototype strain “886-84”. Recently, two additional lineages have been described as possible TBEV subtypes, namely the “strain 178-79”, and the Himalayan subtype (TBEV-HIM)19 (details see chapter 11). So far, it is unclear whether the recently detected strain “Sallandse” from The Netherlands forms an own subtype or belongs to the European subtype.
TBEV-FE prevails in the regions of far-east Russia, in China, Mongolia and in Japan. TBEV-SIB prevails in eastern and western Siberia, in the Ural and European part of Russian territories. TBEV-EU is predominant in Eastern European countries including Ukraine and in central, western, and northern Europe. TBEV-BLK was found in East Siberia near Lake Baikal and in Northern Mongolia, and TBEV-HIM was recently isolated in wild rodent (Marmota himalayana) in the Qinghai-Tibet Plateau in China.20
The principal vector as well as the reservoir for the TBEV-EU subtype is the tick I. ricinus, whereas TBEV-FE and TBEV-SIB subtypes are transmitted predominantly by I. persulcatus. The ranges of the 2 tick species as well as the TBEV subtypes overlap in Estonia, parts of Latvia, Finland, and the European part of Russia.
All 3 main TBEV subtypes are present in Estonia and Latvia.21,22 From the limited virus isolates available from the Ukraine so far, there is evidence that all TBEV subtypes are present on the Crimean peninsula, too.23,24 The TBEV-SIB has been detected in Bosnia as well.23
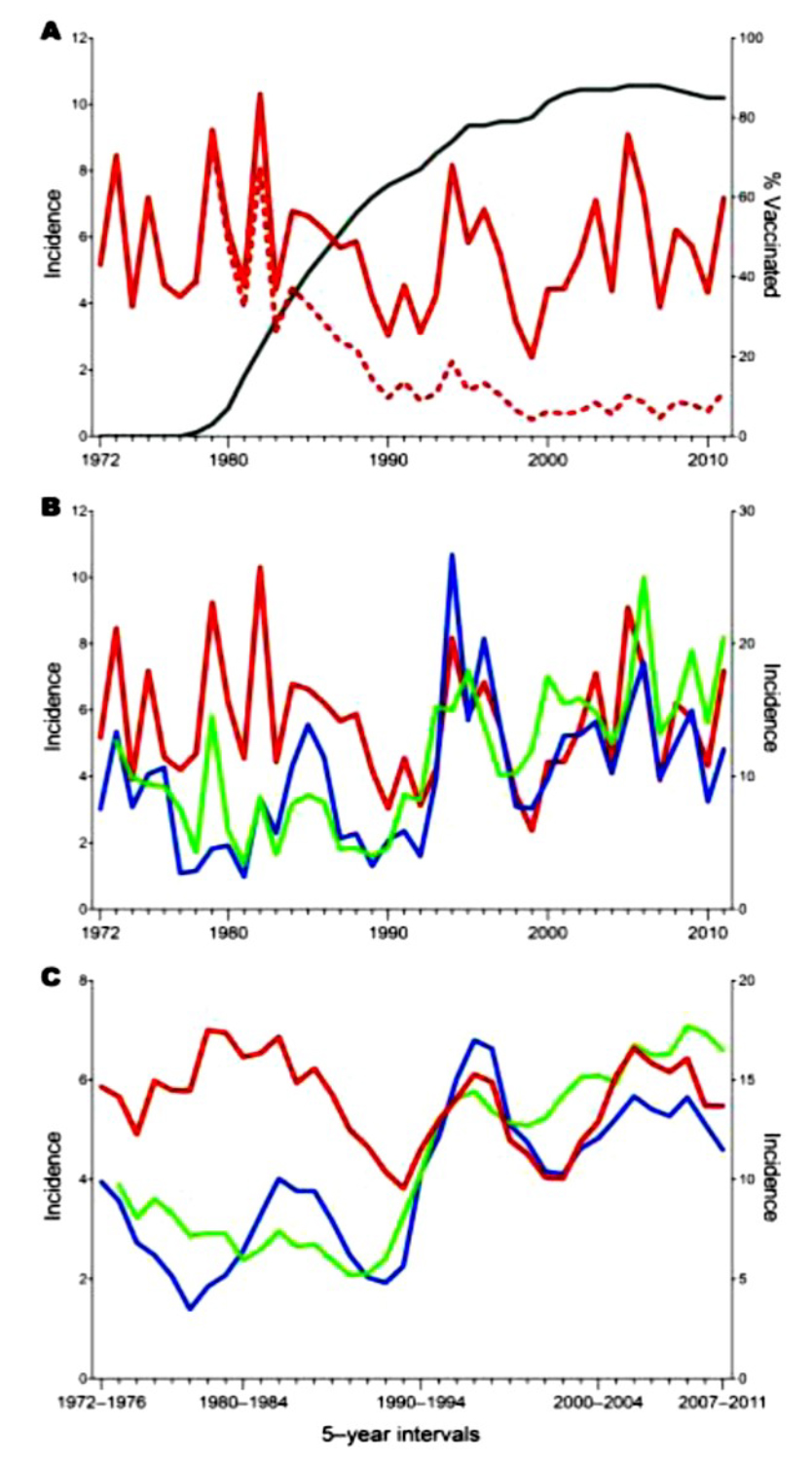
Figure 4: Tick-borne encephalitis (TBE) incidence rates, 1972–2011, Central Europe
Click the image above to enlarge
- Total population (red dashed line) and non-vaccinated population (red solid line) in Austria. The black line represents the increasing coverage of vaccination, which started in 1978.
- Comparative representation of TBE incidences in Austria (red line), Czech Republic (green line), and Slovenia (blue line). The incidence scale for Slovenia (right y-axis) differs from that of Austria and the Czech Republic (left y-axis).
- Sliding-window representation of TBE incidence in Austria (red line), Czech Republic (green line), and Slovenia (blue line) in means of 5-year intervals. The incidence scale for Slovenia (right y-axis) differs from that of Austria and the Czech Republic (left y-axis)1
TBEV-EU foci have been reported from South Korea, approximately 7000 km away from the European range of the TBEV-EU subtype circulation.25 TBEV strains related to the TBEV-EU subtype were isolated in rodents and humans in eastern and western Siberia as well as in the Ural territory.23,26
TBEV-FE foci have not only been reported from Crimea, about 3000 km away from the known TBEV-FE circulation area27 but also from the Republic of Moldova between 2010 and 2011.
Geographical circulation of the TBEV subtypes, unusual TBEV subtype foci, and various carrier vectors are described in more detail in Chapters 3 and 13.
Trends in TBE epidemiology
A characteristic feature of TBE is that the incidence of the disease in risk areas can vary significantly from year to year. In addition to short-term fluctuations, there are also longer-range undulations of incidence rates in intervals of >5 years, which have been analyzed in detail for Austria, the Czech Republic, and Slovenia1 (see Figure 4). Except for the strong overall upsurge of TBE cases in the Czech Republic and Slovenia starting around 1992 (but not in Austria, as a result of vaccination), the long-range incidence curves for 1990–2011 are remarkably similar for all 3 countries, suggesting that the causes for the increase in TBE cases are the same but not yet identified.
A similar fluctuation over time has been recognized in Estonia, a country with one of the highest overall TBE incidence in Europe. Looking into more detail for the years 2005 till 2017 case numbers are fluctuating between 6.2 and 18.6, and when comparing different counties, mean incidence (2005-2017) vary between 5.2 and 52.8 (see Chapter 12b, Estonia).
Again, as noted above, the epidemiology of TBE is a ‘moving target.’ Current changes include an increase in geographical distribution of TBE-risk areas as well as an overall increase of reported TBE cases (Table 2). In recent years new TBE foci have been reported from altitudes up to 2100 meters above sea level,29,30,31,49 New endemic zones in previously unaffected alpine regions in western Austria32 and in Switzerland were established, and a first report of TBEV being detected at locations in Norway up to more than 65.°N latitude was published 2018.33 Within the last couple of years, 4 TBE cases (2 proven and 2 suspected ones) have been identified in UK and Scotland (for more details, see country chapter UK). A remarkable increase in annual disease numbers over the last couple of years is seen in Central European countries, i.e., Austria (most common in unvaccinated subjects), the Czech Republic, Germany, Sweden and Switzerland (see Table 3).
It appears that areas with TBEV endemicity as well as the total number of reported TBE cases have increased over the last several decades. Comparing the periods from 1976 to 1989 and 1990 to 2009, the average increase in TBE infections among humans in central and western European countries was 317.8% in Europe including Russia and it was 193.2% in Europe excluding Russia.34
Various factors may explain these findings, at least in part: social factors (socio-political changes with changes in human behavior, duration and type of leisure time activities), ecological factors (e.g., effects of climate changes on the tick population and reservoir animals), and/ or technological factors (advanced diagnostics, increased medical awareness).
There is increasing research interest in habitat suitability modelling to define universal environmental characteristics of TBEV foci, to predict suitable conditions where potentially human TBEV infections may occur.50-52
Certainly, reporting of TBE cases has improved substantially over the years, and TBE is now a notifiable disease in the EU. In the end, all factors mentioned above play an ‘interactive role’ resulting in complex interactions that may explain the observed changes in TBE epidemiology.
The country reports in Chapter 12b provide standardized information, as available on:
- The history of TBE in the respective country as well as various specific aspects
- Virus, vector, transmission of TBE
- TBE-reporting and prevention by vaccination
- TBE case numbers over time
- Local demographics of TBE
- TBEV-isolation and TBE cases – risk area distribution
Chapter 12c provides a risk map for TBEV based on documented TBE cases, TBEV infection, as well as on the detection of TBEV-circulation in nature (i.e., imperiled, affected and endemic areas). The map does not reflect the incidence of the disease or the universal prevalence of the virus in a given area. As the quality, intensity and completeness of epidemiological surveillance varies between different countries, the map presented here must be incomplete, and very likely TBEV infections and thus TBE may occur in additional (‘new’) areas.
Acknowledgement:
We thank all authors and the co-editors of THE TBE BOOK let no stone unturned in their efforts to find current country-specific information on as many countries as possible and we would like to thank again all authors of Chapter 12b for providing their timely reports.
Contact:
Wilhelm.Erber@pfizer.com
Citation:
Erber W, Schmitt HJ, Janković TV. TBE-epidemiology by country—an overview. Chapter 12a. In: Dobler G, Erber W, Bröker M, Schmitt, HJ, eds. The TBE Book. 6th ed. Singapore: Global Health Press; 2023. doi: 10.33442/26613980_12a-6
References
- Heinz FX, Stiasny K, Holzmann H, et al. Vaccination and tick-borne encephalitis, central Europe. Emerg Infect Dis. 2013;19(1):69-76.
- Commission E. Commission Decision 2002/253/EC of 19 March 2002 laying down case definitions for reporting communicable diseases to the Community network under Decision No 2119/98/EC of the European Parliament and of the Council. EC; 2002. In: EC, ed. Official Journal of the European Union L. 262/12012.
- Kollaritsch H, Krasilnikov V, Holzmann H, et al. Background document on vaccines and vaccination against tick–borne encephalitis. Geneva, WHO Strategic Advisory Group of Experts on Immunization. Available at: http://www.who.int/immunization/sage/6_TBE_backgr_18_Mar_net_apr_2011.pdf (last accessed May 2012). 2011.
- Vaccines against tick-borne encephalitis: WHO position paper. Releve epidemiologique hebdomadaire / Section d’hygiene du Secretariat de la Societe des Nations = Weekly epidemiological record / Health Section of the Secretariat of the League of Nations. 2011;86(24):241-256
- World Health Organization (WHO) ROfEC. Tick-borne encephalitis and haemorrhagic fever with renal syndrome in Europe: report on a WHO meeting Baden 3-5 October 1983. Euro Reports and Studies. 1986;104.
- Donoso Mantke O, Escadafal C, Niedrig M, Pfeffer M, Working Group For Tick-Borne Encephalitis Virus C. Tick-borne encephalitis in Europe, 2007 to 2009. Euro Surveill. 2011;16(39).
- Stefanoff P, Zielicka-Hardy A, Hlebowicz M, et al. New endemic foci of tick-borne encephalitis (TBE) identified in districts where testing for TBE was not available before 2009 in Poland. Parasit Vectors. 2013;6:180.
- Suess J. Tick-borne encephalitis 2010: epidemiology, risk areas, and virus strains in Europe and Asia-an overview. Ticks Tick Borne Dis. 2011;2(1):2-15.
- Suess J. Epidemiology and ecology of TBE relevant to the production of effective vaccines. 2003;21 Suppl 1:S19-35.
- Domanovic D, Giesecke J. How to define an area where transmission of arthropod-borne disease is occurring? Euro Surveill. 2012;17(20).
- Weststrate AC, Knapen D, Laverman GD, et al. Increasing evidence of tick-borne encephalitis (TBE) virus transmission, the Netherlands, June 2016. Euro Surveill. 2017;22(11).
- Dekker M, Laverman GD, de Vries A, Reimerink J, Geeraedts F. Emergence of tick-borne encephalitis (TBE) in the Netherlands. Ticks Tick Borne Dis. 2018.
- Garcia-Bocanegra I, Jurado-Tarifa E, Cano-Terriza D, Martinez R, Perez-Marin JE, Lecollinet S. Exposure to West Nile virus and tick-borne encephalitis virus in dogs in Spain. Transbound Emerg Dis.
- Vanhomwegen J, Beck C, Despres P, et al. Circulation of Zoonotic Arboviruses in Equine Populations of Mallorca Island (Spain). Vector Borne Zoonotic Dis. 2017;17(5):340-346.
- Ergunay K, Saygan MB, Aydogan S, et al. Confirmed exposure to tick-borne encephalitis virus and probable human cases of tick-borne encephalitis in Central/Northern Anatolia, Turkey. Zoonoses Public Health. 2011;58(3):220-227.
- Yilmaz H, Barut K, Karakullukcu A, et al. Serological Evidence of Tick-Borne Encephalitis and West Nile Virus Infections Among Children with Arthritis in Turkey. Vector Borne Zoonotic Dis. 2019.
- Pavlidou V, Geroy S, Diza E, Antoniadis A, Papa A. Epidemiological study of tick-borne encephalitis virus in northern Greece. Vector Borne Zoonotic Dis. 2007;7(4):611-615.
- Pavlidou V, Gerou S, Diza E, Antoniadis A, Papa A. Genetic study of the distribution of Greek goat encephalitis virus in Greece. Vector Borne Zoonotic Dis. 2008;8(3):351-354.
- Pugliese A, Beltramo T, Torre D. Seroprevalence study of Tick-borne encephalitis, Borrelia burgdorferi, Dengue and Toscana virus in Turin Province. Cell Biochem Funct. 2007;25(2):185-188.
- Dai X, Shang G, Lu S, Yang J, Xu J. A new subtype of eastern tick-borne encephalitis virus discovered in Qinghai-Tibet Plateau, China. Emerg Microbes Infect. 2018;7(1):74.
- Lundkvist k, Vene S, Golovljova I, et al. Characterization of tick-borne encephalitis virus from Latvia: evidence for co-circulation of three distinct subtypes. J Med Virol. 2001;65(4):730-735.
- Golovljova I, Vene S, Sjolander KB, Vasilenko V, Plyusnin A, Lundkvist A. Characterization of tick-borne encephalitis virus from Estonia. J Med Virol. 2004;74(4):580-588.
- Tkachev S, et al. Genetic diversity and geographical distribution of the siberian subtype of the tick-borne encephalitis virus. Medical Biodefense Conference 2018; Munich.
- Yurchenko OO, Dubina DO, Vynograd NO, Gonzalez JP. Partial Characterization of Tick-Borne Encephalitis Virus Isolates from Ticks of Southern Ukraine. Vector Borne Zoonotic Dis. 2017.
- Ko S, Kang JG, Kim SY, et al. Prevalence of tick-borne encephalitis virus in ticks from southern Korea. J Vet Sci. 2010;11(3):197-203.
- Adelshin RV, Melnikova OV, Karan LS, Andaev EI, Balakhonov SV. Complete Genome Sequences of Four European Subtype Strains of Tick-Borne Encephalitis Virus from Eastern Siberia, Russia. Genome Announc. 2015;3(3).
- Evstaf’ev IL. [Results of the 20-year study of tick-borne encephalitis in Crimea]. Zh Mikrobiol Epidemiol Immunobiol. 2001(2):111-114.
- Ponomareva EP, Mikryukova TP, Gori AV, et al. Detection of Far-Eastern subtype of tick-borne encephalitis viral RNA in ticks collected in the Republic of Moldova. J Vector Borne Dis. 2015;52(4):334-336.
- Briggs BJ, Atkinson B, Czechowski DM, et al. Tick-borne encephalitis virus, Kyrgyzstan. Emerg Infect Dis. 2011;17(5):876-879.
- Holzmann H, Aberle SW, Stiasny K, et al. Tick-borne encephalitis from eating goat cheese in a mountain region of Austria. Emerg Infect Dis. 2009;15(10):1671-1673.
- Lukan M, Bullova E, Petko B. Climate warming and tick-borne encephalitis, Slovakia. Emerg Infect Dis. 2010;16(3):524-526.
- Heinz FX, Stiasny K, Holzmann H, et al. Emergence of tick-borne encephalitis in new endemic areas in Austria: 42 years of surveillance. Euro Surveill.
- Soleng A, Edgar KS, Paulsen KM, et al. Distribution of ricinus ticks and prevalence of tick-borne encephalitis virus among questing ticks in the Arctic Circle region of northern Norway. Ticks Tick Borne Dis. 2018;9(1):97-103.
- Suess J. Tick-borne encephalitis in Europe and beyond–the epidemiological situation as of 2007. Euro Surveill. 2008;13(26)
- World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO
- Superior Health Council. Vaccination against Tick-Borne Encephalitis (TBE). Brussels: SHC; 2019. Report 9435. https://www.health.belgium.be/sites/default/files/uploads/fields/fpshealth_theme_file/shc_9435_tbe.pdf
- Monthly Newsletter on infectious diseases concerning the news of the current month. 2018. Available from: URL:< https://epidemio.wiv-isp.be/ID/Pages/flashs.aspx>
- Holding M, Dowall SD, Medlock JM, et al. Detection of new endemic focus of tick-borne encephalitis virus (TBEV), Hampshire/Dorset border, England, September 2019. Euro Surveill. 2019;24(47).
- Holding M, Dowall SD, Medlock JM, et al. Tick-Borne Encephalitis Virus, United Kingdom. Emerg Infect Dis. 2020;26(1).
- https://thl.fi/en/web/thlfi-en/-/vaccination-programme-for-tick-borne-encephalitis-expands-to-include-kustavi-and-new-parts-of-the-lake-lohjanjarvi-archipelago
- Salehi-Vaziri M, Pouriayevali MH, Azad-Manjiri S, Ahmadi Vasmehjani A, Baniasadi V, Fazlalipour M. The Seroprevalence of Tick-Borne Encephalitis in Rural Population of Mazandaran Province, Northern Iran (2018 – 2019). Arch Clin Infect Dis. 2020;15(1):e98867.
- European Centre for Disease Prevention and Control. Tick-borne encephalitis. In: ECDC. Annual epidemiological report for 2020. Stockholm: ECDC; 2022.
- Eltari E, Gina A, Bitri T, Sharofi F. Some data on Arboviruses, especially tick-borne encephalitis, in Albania. G Mal Infect Parassit. 1993;45(5):404–11.
- Eltari E. Epidemiology of tick-borne encephalitis in Albania. Ellipse. 1991;29:449–50.
- Burger S. Tick-borne Encephalitis: Global status, 2017 edition. 2017.
- Elyan DS, Moustafa L, Noormal B, et al. Serological evidence of flaviviruses infection among acute febrile illness patients in Afghanistan. J Infect Dev Ctries. 2014;8(9):1176–80.
- Kuchuloria T, Imnadze P, Mamuchishvili N, et al. Hospital-based surveillance for infectious etiologies among patients with acute febrile illness in Georgia, 2008-2011. Am J Trop Med Hyg. 2016;94(1):236–42.
- Atkinson B, Hewson R. Emerging arboviruses of clinical importance in Central Asia. J Gen Virol. 2018;99(9):1172-1184.
- Daniel M, Danielová V, Kríz B, Jirsa A, Nozicka J. Shift of the tick Ixodes ricinus and tick-borne encephalitis to higher altitudes in central Europe. Eur J Clin Microbiol Infect Dis. 2003;22(5):327-328.
- WHO 2018. World health report of an international consultation: methodology for risk mapping of the international spread of vector-borne diseases via air travel: Geneva, Switzerland, 5-6 July 2018.
- Borde JP, Glaser R, Braun K, et al. Decoding the Geography of Natural TBEV Microfoci in Germany: A Geostatistical Approach Based on Land-Use Patterns and Climatological Conditions. Int J Environ Res Public Health. 2022;19(18):11830.
- Gray JS, Dautel H, Estrada-Peña A, Kahl O, Lindgren E. Effects of climate change on ticks and tick-borne diseases in Europe. Interdiscip Perspect Infect Dis. 2009;2009:593232.